��Ŀ����
(8��)�����£���������������Ӧ��
a��0.1mol/L������Һ��CH3COOH  CH3COO-��H+
CH3COO-��H+
b��0.1mol/L��������Һ��CH3COO-��H2O  CH3COOH��OH-
CH3COOH��OH-
(1)��ʽ����ƽ�����a�м���һ������ˮ��ƽ����________________�ƶ�����Һ��pH______(����ߡ������͡������䡱)
(2)��b�м���ͬŨ�ȵĴ�������Һ����Һ��pH��_______(����ߡ������͡������䡱)��������������CH3COOH��ʹ��Һ��c(Na+)��c(CH 3COO-)������Һ��pHΪ_______
3COO-)������Һ��pHΪ_______ (����ֵ)��
(����ֵ)��
(1)��(��������)������ (2)���䡢7
����

��ϰ��ϵ�д�
�����Ŀ
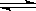 CH3COO-��H+
CH3COO-��H+