��Ŀ����
ij�о�С���Ի�������Ͳ����Ϊ��Ҫԭ�ϣ�������·�ߺϳ�ҩ�ﲼ������ʰ����ơ�

��֪����1��RCOOH  RCOC1
RCOC1  RCOOR�@
RCOOR�@
��2��R-Cl-  R-NH2
R-NH2  R-NH2HC1
R-NH2HC1 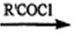 RCONHR
RCONHR
��ش�
��1��д��������Ľṹ��ʽ��B_________ �� D_________ ��
��2������˵������ȷ����_________��
A�� ת��ΪAΪ������Ӧ
ת��ΪAΪ������Ӧ
B�� RCOOH��SOCl2��Ӧ�IJ�����SO2��HC1
C��������B�ܷ������۷�Ӧ
D����������ʰ����Ƶķ���ʽΪC15H19NO3Na
��3��д��ͬʱ�������������IJ���ҵ�����ͬ���칹��_________��
�ٺ�����ױ��������к���������ʵ�鷢������NaOH��Һ1��2��Ӧ��Ҳ�ܷ���������Ӧ��
��H��NMR����ʾ��������������ͬ�����ұ�����ֻ��һ�ֻ�ѧ��������ԭ�ӡ�
��4��д��F����������ʰ����ƵĻ�ѧ����ʽ_________��
��5�����������Ϣ������� Ϊԭ���Ʊ���
Ϊԭ���Ʊ��� ���ĺϳ�·��(������ͼ��ʾ�����Լ���ѡ)_____________________��
���ĺϳ�·��(������ͼ��ʾ�����Լ���ѡ)_____________________��
���ݱ��е�����,�ӵ縺�ԵĽǶ��ж�����Ԫ��֮�����γɹ��ۼ���һ����(����)
Ԫ�� | Na | Mg | Al | H | C | O | Cl |
�縺�� | 0.9 | 1.2 | 1.5 | 2.1 | 2.5 | 3.5 | 3.0 |
��Na��Cl����Mg��Cl����Al��Cl����H��O����Al��O����C��Cl
A. �٢ڢ� B. �ۢܢ� C. �ܢݢ� D. ȫ��
�������ʵķ�����ȷ���� �� ��
�� | �� | �� | ���������� | ���������� | |
A | �ռ� | H2SO4 | BaCO3 | Na2O2 | CO2 |
B | Ba(OH)2 | HCl | NaCl | Na2O | CO |
C | ���� | CH3COOH | CaCl2 | Al2O3 | SO2 |
D | KOH | HClO | CaCO3 | CaO | SiO2 |
A. A B. B C. C D. D

 ��
��  ���Ƿ�������
���Ƿ������� ���Ƿ��������Ƿ��㻯����
���Ƿ��������Ƿ��㻯���� ��
�� ����������һ����CH2���������ͬϵ���ϵ
����������һ����CH2���������ͬϵ���ϵ
 ��
�� ��
�� ���ֺ��ؿ����3��ˮ����
���ֺ��ؿ����3��ˮ����