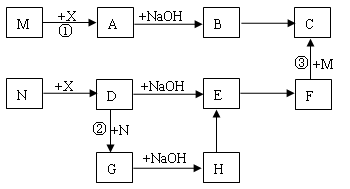
��Ŀ����
C��D��G��I��Ϊ������Ԫ���γɵĵ��ʣ�D��G��IΪ�����ǽ�����̬���ʡ�DԪ�ص�ԭ�ӵ������������Ǵ�����������3����C��Gͬ���ڣ���ԭ���������������4�����ǵļ����ӵ��Ӳ�ṹ��ͬ�����������ת����ϵ��

����գ�
(1) D��I���γ� ԭ�Ӹ�����Ϊ1��1�Ĺ��ۻ������д�������ʽ�� ��
ԭ�Ӹ�����Ϊ1��1�Ĺ��ۻ������д�������ʽ�� ��
��2��L��ĿǰӦ����㷺�Ľ�������̼����������L��������д�����Eˮ��Һ�Ļ�ѧʽ�� ��
��3��������K �к�����ɵ���L��Ԫ�أ��Ҹ�Ԫ�ص���������Ϊ70%����Ӧ�ٵĻ�ѧ����ʽ�� �������÷�Ӧ�IJ�����
��4��д��A+F �� J�����ӷ���ʽ�� ��

����գ�
(1) D��I���γ�
 ԭ�Ӹ�����Ϊ1��1�Ĺ��ۻ������д�������ʽ�� ��
ԭ�Ӹ�����Ϊ1��1�Ĺ��ۻ������д�������ʽ�� ����2��L��ĿǰӦ����㷺�Ľ�������̼����������L��������д�����Eˮ��Һ�Ļ�ѧʽ�� ��
��3��������K �к�����ɵ���L��Ԫ�أ��Ҹ�Ԫ�ص���������Ϊ70%����Ӧ�ٵĻ�ѧ����ʽ�� �������÷�Ӧ�IJ�����
��4��д��A+F �� J�����ӷ���ʽ�� ��
��1��H2O2��2�֣�
(2) 2KCl+2H2O 2KOH+ H
2KOH+ H 2��+Cl2�� ��3�֣�
2��+Cl2�� ��3�֣�
(3) Fe2O3 +2Al 2Fe + Al2O3��3�֣�
2Fe + Al2O3��3�֣�
�ڻ�����ϼ�����KClO3������Mg���������ȼ��3�֣�
(4)Al2O3+2OH-===2AlO2-+H2O ��3�֣�
(2) 2KCl+2H2O
 2KOH+ H
2KOH+ H 2��+Cl2�� ��3�֣�
2��+Cl2�� ��3�֣�(3) Fe2O3 +2Al
 2Fe + Al2O3��3�֣�
2Fe + Al2O3��3�֣��ڻ�����ϼ�����KClO3������Mg���������ȼ��3�֣�
(4)Al2O3+2OH-===2AlO2-+H2O ��3�֣�
��

��ϰ��ϵ�д�
 ���������ּ���ÿһ��ȫ�º�����ҵ��ϵ�д�
���������ּ���ÿһ��ȫ�º�����ҵ��ϵ�д� ��ٽ������½������������ϵ�д�
��ٽ������½������������ϵ�д�
�����Ŀ
 2������X�����Ԫ��ԭ�ӽṹ��ͼ �� ��
2������X�����Ԫ��ԭ�ӽṹ��ͼ �� ��