��Ŀ����
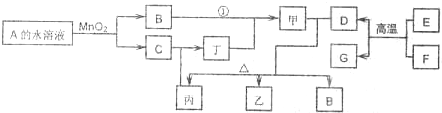
��1��A�ĽṹʽΪ
��2������Ӧ�ٳ����ɼ��⣬�����ɱ���д������D���ϡ��Һ��Ӧʱ�����ӷ���ʽ��
��3����D��FΪ�缫��NaOH��ҺΪ�������Һ�������ԭ��أ���ԭ��ظ����ĵ缫��ӦʽΪ
��4����A������Ի������ɼף���ϡ��Һ��Ũ��Һ�ֱ���D��һ�������·�Ӧ��������������Ԫ����ȫ��ͬ������������������ʣ���д��A����������ɼĻ�ѧ��Ӧ����ʽ��
��1��AΪH2O2���ṹʽΪH-O-O-H��E��D�ķ�Ӧ�����ȷ�Ӧ�������ں��������ұ��������
�ʴ�Ϊ��H-O-O-H�����������ұ��������
��2������D���ϡ��Һ��Ӧʱ�������������������ӷ���ʽΪ3Fe+8H++2NO3-�T3Fe2++2NO��+4H2O���ʴ�Ϊ��3Fe+8H++2NO3-�T3Fe2++2NO��+4H2O��
��3����D��FΪ�缫��NaOH��ҺΪ�������Һ�������ԭ��أ���ط�ӦΪ4Al+3O2+4NaOH=4NaAlO2+2H2O����AlΪ�������缫��ӦΪAl-3e-+4OH-=4AlO2-+2H2O����װ�õ��CuSO4��Һ���ö��Ե缫�����������������ӷŵ������������缫��ӦΪ4OH--4e-=O2��+2H2O���������������������ڱ�״���¶���2.24L��������
| 2.24L |
| 22.4L/mol |
| 1 |
| 2 |
| 0.1mol |
| 0.2L |
�ʴ�Ϊ��Al-3e-+4OH-=4AlO2-+2H2O��4OH--4e-=O2��+2H2O��0.5mol/L��
��4��A����������ɼĻ�ѧ��Ӧ����ʽΪH2O2+SO2=H2SO4��[AlFe��OH��4Cl2]m�����Ũ���ᷴӦ�Ļ�ѧ����ʽΪ[AlFe��OH��4Cl2]m+4mHCl=mAlCl3+mFeCl3+4mH2O���ʴ�Ϊ��H2O2+SO2=H2SO4��[AlFe��OH��4Cl2]m+4mHCl=mAlCl3+mFeCl3+4mH2O��

 ����˼ά����ѵ����ʱ��ѧ��ϵ�д�
����˼ά����ѵ����ʱ��ѧ��ϵ�д�(7��) (1)��ѧƽ�ⳣ��K��ʾ���淴Ӧ�Ľ��г̶ȣ�KֵԽ��ʾ_________��Kֵ��С���¶ȵĹ�ϵ�ǣ��¶����ߣ�Kֵ________(������С����������Ҳ���ܼ�С)��
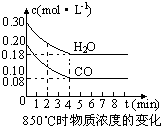
��2����һ���Ϊ10L�������У�ͨ��һ������CO��H2O����850��ʱ�������·�Ӧ��CO(g)ʮH2O(g) CO2(g)ʮH2 (g) ��H��0��CO��H2OŨ�ȱ仯������ͼ���� 0��4min��ƽ����Ӧ����v(CO)��______mol��(L��min)
t1��ʱ����Ũ�ȣ�mol/L���ı仯
| ʱ��(min) | CO | H2O | CO2 | H2 |
| 0 | 0.200 | 0.300 | 0 | 0 |
| 2 | 0.138 | 0.238 | 0.062 | 0.062 |
| 3 | C1 | C2 | C3 | C3 |
| 4 | C1 | C2 | C3 | C3 |
| 5 | 0.116 | 0.216 | 0.084 |
|
| 6 | 0.096 | 0.266 | 0.104 |
|
��3)t��(����850��)ʱ������ͬ�����з���������Ӧ�������ڸ����ʵ�Ũ�ȱ仯���������
�ٱ���3min��4min֮�䷴Ӧ����_________״̬�� C1��ֵ_________0.08 mol��L (����ڡ�С�ڻ����)��
�ڷ�Ӧ��4min��5min֮�䣬ƽ�����淽���ƶ������ܵ�ԭ����________(��ѡ)������5min��6min֮����ֵ�����仯�����ܵ�ԭ����__________(��ѡ)��
a������ˮ���� b�������¶� c��ʹ�ô��� d����������Ũ��
(7��) (1)��ѧƽ�ⳣ��K��ʾ���淴Ӧ�Ľ��г̶ȣ�KֵԽ��ʾ_________��Kֵ��С���¶ȵĹ�ϵ�ǣ��¶����ߣ�Kֵ________(������С����������Ҳ���ܼ�С)��
��2����һ���Ϊ10L�������У�ͨ��һ������CO��H2O����850��ʱ�������·�Ӧ��CO(g)ʮH2O(g)
 CO2(g)ʮH2 (g) ��H��0��CO��H2OŨ�ȱ仯������ͼ���� 0��4min��ƽ����Ӧ����v(CO)��______
mol��(L��min)
CO2(g)ʮH2 (g) ��H��0��CO��H2OŨ�ȱ仯������ͼ���� 0��4min��ƽ����Ӧ����v(CO)��______
mol��(L��min)

t1��ʱ����Ũ�ȣ�mol/L���ı仯
|
ʱ��(min) |
CO |
H2O |
CO2 |
H2 |
|
0 |
0.200 |
0.300 |
0 |
0 |
|
2 |
0.138 |
0.238 |
0.062 |
0.062 |
|
3 |
C1 |
C2 |
C3 |
C3 |
|
4 |
C1 |
C2 |
C3 |
C3 |
|
5 |
0.116 |
0.216 |
0.084 |
|
|
6 |
0.096 |
0.266 |
0.104 |
|
��3)t��(����850��)ʱ������ͬ�����з���������Ӧ�������ڸ����ʵ�Ũ�ȱ仯���������
�ٱ���3min��4min֮�䷴Ӧ����_________״̬�� C1��ֵ_________0.08 mol��L (����ڡ�С�ڻ����)��
�ڷ�Ӧ��4min��5min֮�䣬ƽ�����淽���ƶ������ܵ�ԭ����________(��ѡ)������5min��6min֮����ֵ�����仯�����ܵ�ԭ����__________(��ѡ)��
a������ˮ���� b�������¶� c��ʹ�ô��� d����������Ũ��
 ��1����ѧƽ�ⳣ��K��ʾ���淴Ӧ�Ľ��г̶ȣ�KֵԽ��ʾ
��1����ѧƽ�ⳣ��K��ʾ���淴Ӧ�Ľ��г̶ȣ�KֵԽ��ʾ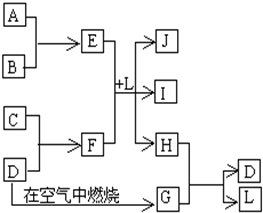 ��ͼÿһ�����е���ĸ����һ�ַ�Ӧ������������J�Ǻ�����Ԫ��A�İ�ɫ��״������IΪNaCl��Һ��D�ǵ���ɫ���嵥�ʣ�����д���пհף�
��ͼÿһ�����е���ĸ����һ�ַ�Ӧ������������J�Ǻ�����Ԫ��A�İ�ɫ��״������IΪNaCl��Һ��D�ǵ���ɫ���嵥�ʣ�����д���пհף�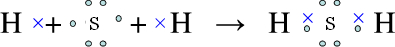
 HS-+OH-
HS-+OH-