��Ŀ����
CuSO4��Cu(NO3)2����Ȼ������Ҫ��ͭ�Ρ��ش��������⣺
��1��CuSO4��Cu(NO3)2�������ӻ�̬��������Ų�ʽΪ_________��S��O��N����Ԫ�صĵ�һ�������ɴ�С��˳��Ϊ_________��
��2����Cu(NO3)2��Һ��ͨ������NH3�����������[Cu(NH3)4] (NO3)2������NO3-����ԭ�ӵ��ӻ��������Ϊ_________��[Cu(NH3)4]NO3)2�д��ڵĻ�ѧ�����ͳ��˼��Թ��ۼ��⣬����_________��
��3��������ͭ��Һ�м������KCN������������[Cu(CN)4]2-,CN-���ṩ�µ��ӶԵ�ԭ����_________�� lmol CN-�к��еĦм�����ĿΪ_________����CN-��Ϊ�ȵ������������_________ ��д��һ�ּ��ɣ�
��4��CuSO4���۵�Ϊ560��C��,Cu(NO3)2���ܵ�Ϊ115��C,CuSO4�ܵ���ߵ�ԭ����_________��
��5������CuSO4��NaOH�Ʊ���Cu(OH)2����ȩ��ʱ�����ɺ�ɫ��Cu2O ,�侧���ṹ��ͼ��ʾ��

�þ���ԭ���������AΪ��0��0��0)��BΪ��1��0��0) ��CΪ�� ��
�� ��
�� ������Dԭ�ӵ��������Ϊ_________��������_________ԭ��(��Ԫ�ط���)��
������Dԭ�ӵ��������Ϊ_________��������_________ԭ��(��Ԫ�ط���)��
����֪����ͭ�Ķѻ���ʽ�������������ܶѻ���������ͭԭ�ӵ���λ����_________���þ�����Cuԭ�ӵĿռ���������_________��
 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�������ͼ��ʾװ�ý�������ʵ�飬�ܵó���Ӧʵ����۵��ǣ� ��
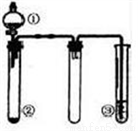
ѡ�� | �� | �� | �� | ʵ����� |
A | ϡ���� | CaCO3 | Na2SiO3��Һ | �ǽ�����:Cl>C>Si |
B | Ũ���� | ���� | Ba(NO3)2��Һ | ��֤SO2������Ա��ο����ɰ�ɫ���� |
C | Ũ��ˮ | ��ʯ�� | ��̪��Һ | ������ˮ��Һ�ʼ��� |
D | Ũ���� | Fe | NaOH��Һ | ����Ũ���ᷴӦ������NO2 |
A. A B. B C. C D. D






