��Ŀ����
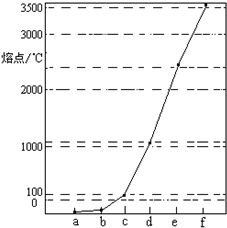 ��ͼ��Na��Cu��Si��H��C��N��Ԫ�ص��ʵ��۵�ߵ͵�˳������c��d�����Ⱥ͵�������壮�����жϲ���ȷ���ǣ�������
��ͼ��Na��Cu��Si��H��C��N��Ԫ�ص��ʵ��۵�ߵ͵�˳������c��d�����Ⱥ͵�������壮�����жϲ���ȷ���ǣ�������������Na��Cu��Si��H��C��N��Ԫ�ص����У�Na��CuΪ�������壬�����Ⱥ͵�������壬C��Si�ĵ���Ϊԭ�Ӿ��壬��C���ʵ��۷е����Siԭ�Ӿ�����۷е㣬H��N��Ӧ�ĵ���Ϊ���Ӿ��壬�����������۵���ͣ���ͼ�۵�ĸߵ�˳���֪aΪH��bΪN��cΪNa��dΪCu��eΪSi��fΪC����϶�Ӧ���ʡ�������Ľṹ�������Լ�Ԫ�������ɵĵݱ���ɽ����⣮
����⣺Na��Cu��Si��H��C��N��Ԫ�ص����У�Na��CuΪ�������壬�����Ⱥ͵�������壬C��Si�ĵ���Ϊԭ�Ӿ��壬��C���ʵ��۷е����Siԭ�Ӿ�����۷е㣬H��N��Ӧ�ĵ���Ϊ���Ӿ��壬�����������۵���ͣ���ͼ�۵�ĸߵ�˳���֪aΪH��bΪN��cΪNa��dΪCu��eΪSi��fΪC��
A��eΪSi��fΪC����Ӧ�ĵ���Ϊԭ�Ӿ��壬���ڹ��ۼ�����A��ȷ��
B��dΪCu����ӦԪ��ԭ�ӵĵ����Ų�ʽ��1s22s22p63s23p63d104s1����B����
C��bΪN��Ԫ���γɵ���ۺ������к���O-H��������ˮ����֮���γ��������C��ȷ��
D������a��b��f��Ӧ��Ԫ����ԭ�Ӹ�����1��1��1�γɵķ���ΪHCN���ṹʽΪH-C��N�������к�2���Ҽ���2���м�����D��ȷ��
��ѡB��
A��eΪSi��fΪC����Ӧ�ĵ���Ϊԭ�Ӿ��壬���ڹ��ۼ�����A��ȷ��
B��dΪCu����ӦԪ��ԭ�ӵĵ����Ų�ʽ��1s22s22p63s23p63d104s1����B����
C��bΪN��Ԫ���γɵ���ۺ������к���O-H��������ˮ����֮���γ��������C��ȷ��
D������a��b��f��Ӧ��Ԫ����ԭ�Ӹ�����1��1��1�γɵķ���ΪHCN���ṹʽΪH-C��N�������к�2���Ҽ���2���м�����D��ȷ��
��ѡB��
���������⿼��Ԫ�ص��ƶϺ;�������ʣ���Ŀ�Ѷ��еȣ����ݾ�����۷е㵽���ƶ�Ԫ�ص�����Ϊ������Ĺؼ���ע����ճ�����������ʺ��ɣ�

��ϰ��ϵ�д�
�����Ŀ
���и����������α�ʾΪa��b��c��d��ͼ�м�ͷ��ʾһ��ת����������ͼת����ϵ���ǣ�������
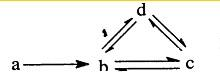
| A��Na Na2O2 NaOH Na2CO3 | B��S SO2 H2SO3 NaHSO3 | C��Cu CuO Cu��OH��2 CuSO4 | D��Si SiO2 H2SiO3 Na2SiO3 |
 ��1��0.1mol/LNa2CO3��Һ��
��1��0.1mol/LNa2CO3��Һ��