��Ŀ����
ij��ѧ��ȤС��ⶨijFe2��SO4��3��Ʒ��ֻ������Fec12���ʣ�����Ԫ�ص�����������������ʵ�鲽����в�����
�ٳ�ȡag��Ʒ�������ձ��У�
�ڼ���50mL 1.0mol/Lϡ�����һ����������ˮ��ʹ��Ʒ�ܽ⣬Ȼ��ȷ���Ƴ�250.00mL��Һ��
����ȡ25.00mL���� ������õ���Һ�������ձ��У�������������ˮ��ʹ��Ӧ��ȫ��
�ܼ��������ˮ����ֽ��裬ʹ������ȫ��
�ݹ��ˣ�ϴ�ӳ�����
������ת�Ƶ�ij�����ڣ����ȡ����裬ֱ�������ɺ��ɫȫ����Ϊ����ɫ���ڸ���������ȴ�����º�����
�ߡ�
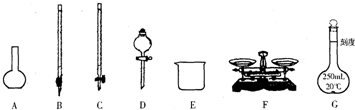
����������������ش�
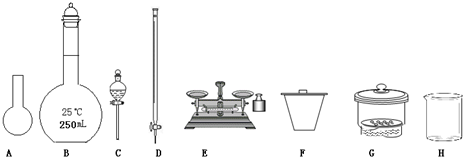
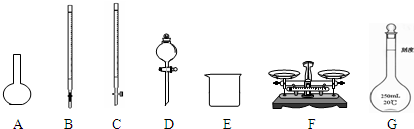
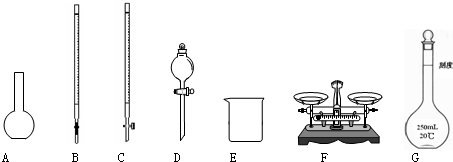
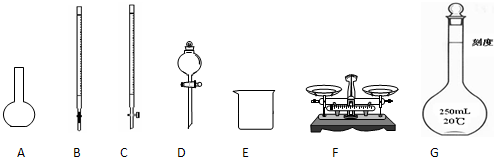
��1����ͼ��ʾ�����У���ʵ�鲽��٢ڢ��б����õ���������E�� ������ĸ����
��2��������У�����50mL��1.0mol/LϡH2SO4��Ҫ98%���ܶ�1.84g/cm3����ŨH2SO4���Ϊ mL����ȡ�������ŨH2SO4�õ���Ͳ����� ��
��3����Ʒ�е�����Fe2+�н�ǿ�Ļ�ԭ�ԣ���ɲ���ƽ���з�Ӧ�����ӷ���ʽ
Fe2++ ClO2+ �T Fe3++ Cl-+ H2O
�������Ϸ���������ʵ����������ˮ��ΪClO2ʱ����ʵ������� ����ƫ��ƫС��Ӱ�죩�������ʵ�����ClO2��Cl2������Ч��֮��Ϊ ��
��4���ڢ��IJ����У���������ת�Ƶ� �����������ƣ��м��ȣ���ȴ�����£�����������Ϊm1g���ٴμ��Ȳ���ȴ�����³���������Ϊm2g����m1��m2��ֵ�ϴ������IJ���Ӧ���� ��
��5����������ڸ���������ȴ����ⶨ����Ԫ�ص����������� ����ƫ��ƫС��Ӱ�죩��������������W1g�����������������������W2g������Ʒ����Ԫ�ص���������Ϊ �����г���ʽ�����軯��
�ٳ�ȡag��Ʒ�������ձ��У�
�ڼ���50mL 1.0mol/Lϡ�����һ����������ˮ��ʹ��Ʒ�ܽ⣬Ȼ��ȷ���Ƴ�250.00mL��Һ��
����ȡ25.00mL���� ������õ���Һ�������ձ��У�������������ˮ��ʹ��Ӧ��ȫ��
�ܼ��������ˮ����ֽ��裬ʹ������ȫ��
�ݹ��ˣ�ϴ�ӳ�����
������ת�Ƶ�ij�����ڣ����ȡ����裬ֱ�������ɺ��ɫȫ����Ϊ����ɫ���ڸ���������ȴ�����º�����
�ߡ�

����������������ش�
��1����ͼ��ʾ�����У���ʵ�鲽��٢ڢ��б����õ���������E��
��2��������У�����50mL��1.0mol/LϡH2SO4��Ҫ98%���ܶ�1.84g/cm3����ŨH2SO4���Ϊ
��3����Ʒ�е�����Fe2+�н�ǿ�Ļ�ԭ�ԣ���ɲ���ƽ���з�Ӧ�����ӷ���ʽ
�������Ϸ���������ʵ����������ˮ��ΪClO2ʱ����ʵ�������
��4���ڢ��IJ����У���������ת�Ƶ�
��5����������ڸ���������ȴ����ⶨ����Ԫ�ص�����������
��������1�����ݸ�����������ѡȡ������
��2������������Һϡ���������ʵ����������������Һ�����������Һ�����Сѡ����Ͳ��
��3������������ԭ��Ӧ�����غ㡢ԭ���غ���ƽ���ӷ���ʽ����ԭ��һ������Ҫ�������������������������ƣ���ԭ��ʧ������һ�����������ı䲻Ӱ������������������������ǵõ�������֮�ȣ�
��4����������ֱ�������ɺ��ɫȫ����Ϊ����ɫ����������������н��У�������������صı������γ�������������0.1g��
��5������ʵ�鲽�������֪����������Ϊ������Ʒ�����������������������������ͳ����������ٽ������γ��������أ����ڹ����г���������Ԫ�������غ㣬������ɫ���壨Fe2O3���е���������Ʒ�������������������Ĺ�ʽ�����Ԫ�ص�����������
��2������������Һϡ���������ʵ����������������Һ�����������Һ�����Сѡ����Ͳ��
��3������������ԭ��Ӧ�����غ㡢ԭ���غ���ƽ���ӷ���ʽ����ԭ��һ������Ҫ�������������������������ƣ���ԭ��ʧ������һ�����������ı䲻Ӱ������������������������ǵõ�������֮�ȣ�
��4����������ֱ�������ɺ��ɫȫ����Ϊ����ɫ����������������н��У�������������صı������γ�������������0.1g��
��5������ʵ�鲽�������֪����������Ϊ������Ʒ�����������������������������ͳ����������ٽ������γ��������أ����ڹ����г���������Ԫ�������غ㣬������ɫ���壨Fe2O3���е���������Ʒ�������������������Ĺ�ʽ�����Ԫ�ص�����������
����⣺��1������ҩƷ����ƽ���Ȼ������Ȼ�������ˮ��Һ�������ԣ�����ȷ��ȡ25.00mL���������õ���Һ����ʽ�ζ��ܣ�����һ�����ʵ���Ũ�ȵ���Һ������ƿ��
�ʴ�Ϊ��CFG��
��2������50mL��1.0mol/LϡH2SO4��Ҫ98%���ܶ�1.84g/cm3����ŨH2SO4���Ϊ��ΪVml������ϡ��ǰ����Һ�������ʵ������䣬
=0.05L��1.0mol/L��V=2.7ml����ȡ�������ŨH2SO4�õ���Ͳ�����10ml��
�ʴ�Ϊ��2.7��10ml��
��3����Ӧ�У��������ӱ仯Ϊ�����ӣ������������Ԫ�ػ��ϼ۴�+4�۱仯Ϊ-1�ۣ��仯5�ۣ�����ת����С������Ϊ5�����ݵ����غ��ԭ���غ���ƽ�õ����ӷ���ʽΪ��5Fe2++ClO2+4 H+�T5Fe3++Cl-+2H2O���������Ϸ���������ʵ����������ˮ��ΪClO2ʱ��ʵ����Ӱ�죬��ͬ������������ͬ��ͬ����ԭ�����ʱ�������Ҫʧȥ������ͬ�������ʵ�����ClO2��Cl2������Ч��֮��ΪΪת�Ƶ�����֮�ȣ�ClO2��Cl-��5e-��Cl2��2Cl-��2e-������1molClO2��Cl2������Ч��֮��Ϊ5��2��
�ʴ�Ϊ��5��1��4��H+��5��1��2����Ӱ�죻5��2��
��4��������ת�Ƶ������ڣ����ȡ����裬ֱ�������ɺ��ɫȫ����Ϊ����ɫ���ڸ���������ȴ�����º����ƺ��أ����γ�����ֵ������0.1g��
�ʴ�Ϊ����������������һ��ʱ������������ڸ���������ȴ���ٳ�������������γƵõ����������0.1gΪֹ��
��5����������ڸ���������ȴ�������տ����е�ˮ����������������������ⶨ����Ԫ�ص���������ƫ�ߣ�������������W1g�����������������������W2g��ע��ʵ��ⶨ25ml��Һ������������������Ʒ����Ԫ����250ml��Һ��������Ԫ�ص�����������������Ԫ����������=
��2��56g/mol��
��ag��100%��
�ʴ�Ϊ������
��2��56g/mol��
��ag��100%��
�ʴ�Ϊ��CFG��
��2������50mL��1.0mol/LϡH2SO4��Ҫ98%���ܶ�1.84g/cm3����ŨH2SO4���Ϊ��ΪVml������ϡ��ǰ����Һ�������ʵ������䣬
| Vml��1.84g/ml��98% |
| 98g/mol |
�ʴ�Ϊ��2.7��10ml��
��3����Ӧ�У��������ӱ仯Ϊ�����ӣ������������Ԫ�ػ��ϼ۴�+4�۱仯Ϊ-1�ۣ��仯5�ۣ�����ת����С������Ϊ5�����ݵ����غ��ԭ���غ���ƽ�õ����ӷ���ʽΪ��5Fe2++ClO2+4 H+�T5Fe3++Cl-+2H2O���������Ϸ���������ʵ����������ˮ��ΪClO2ʱ��ʵ����Ӱ�죬��ͬ������������ͬ��ͬ����ԭ�����ʱ�������Ҫʧȥ������ͬ�������ʵ�����ClO2��Cl2������Ч��֮��ΪΪת�Ƶ�����֮�ȣ�ClO2��Cl-��5e-��Cl2��2Cl-��2e-������1molClO2��Cl2������Ч��֮��Ϊ5��2��
�ʴ�Ϊ��5��1��4��H+��5��1��2����Ӱ�죻5��2��
��4��������ת�Ƶ������ڣ����ȡ����裬ֱ�������ɺ��ɫȫ����Ϊ����ɫ���ڸ���������ȴ�����º����ƺ��أ����γ�����ֵ������0.1g��
�ʴ�Ϊ����������������һ��ʱ������������ڸ���������ȴ���ٳ�������������γƵõ����������0.1gΪֹ��
��5����������ڸ���������ȴ�������տ����е�ˮ����������������������ⶨ����Ԫ�ص���������ƫ�ߣ�������������W1g�����������������������W2g��ע��ʵ��ⶨ25ml��Һ������������������Ʒ����Ԫ����250ml��Һ��������Ԫ�ص�����������������Ԫ����������=
| (W2-W1)g |
| 160g/mol |
| 250mL |
| 25mL |
�ʴ�Ϊ������
| (W2-W1)g |
| 160g/mol |
| 250mL |
| 25mL |
���������⿼����ʵ��̽��������ʵ��ⶨ����Һ���ƹ��̣���������������������ԭ��Ӧ��ƽ�ǽ���ؼ���ע��ʵ��ⶨ��Һ����ı仯����Ŀ�Ѷ��еȣ�

��ϰ��ϵ�д�
�����Ŀ