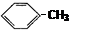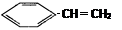ΧβΡΩΡΎ»ί
Ρ≥ΧΰA 0.2 mol ‘Ύ―θΤχ÷–Άξ»Ϊ»Φ…’ΚσΘ§…ζ≥…CO2ΚΆH2OΗς1.2 molΓΘ ‘ΜΊ¥πΘΚ
Θ®1Θ©ΧΰAΒΡΖ÷Ή” ΫΈΣ_____________ΓΘ
Θ®2Θ©»τ»Γ“ΜΕ®ΝΩΒΡΧΰAΆξ»Ϊ»Φ…’ΚσΘ§…ζ≥…CO2ΚΆH2OΗς3 molΘ§‘ρ”–________gΧΰA≤ΈΦ”ΝΥΖ¥”ΠΘ§»Φ…’ ±œϊΚΡ±ξΉΦΉ¥Ωωœ¬ΒΡ―θΤχ___________LΓΘ
Θ®3Θ©»τΧΰA≤ΜΡή ΙδεΥ°Ά …ΪΘ§ΒΪ‘Ύ“ΜΕ®ΧθΦΰœ¬ΡޔꬻΤχΖΔ…ζ»Γ¥ζΖ¥”ΠΘ§Τδ“Μ¬»»Γ¥ζΈο÷Μ”–“Μ÷÷Θ§‘ρΧΰAΒΡΫαΙΙΦρ ΫΈΣ__________________ΓΘ
Θ®4Θ©»τΧΰAΡή ΙδεΥ°Ά …ΪΘ§‘Ύ¥ΏΜ·ΦΝΉς”Οœ¬Θ§”κH2Φ”≥…Θ§ΤδΦ”≥…≤ζΈοΨ≠≤βΕ®Ζ÷Ή”÷–Κ§”–4ΗωΦΉΜυΘ§ΧΰAΩ…Ρή”–ΒΡΫαΙΙΦρΦρ Ϋ__________________________________________ΓΘ
Θ®1Θ©ΧΰAΒΡΖ÷Ή” ΫΈΣ_____________ΓΘ
Θ®2Θ©»τ»Γ“ΜΕ®ΝΩΒΡΧΰAΆξ»Ϊ»Φ…’ΚσΘ§…ζ≥…CO2ΚΆH2OΗς3 molΘ§‘ρ”–________gΧΰA≤ΈΦ”ΝΥΖ¥”ΠΘ§»Φ…’ ±œϊΚΡ±ξΉΦΉ¥Ωωœ¬ΒΡ―θΤχ___________LΓΘ
Θ®3Θ©»τΧΰA≤ΜΡή ΙδεΥ°Ά …ΪΘ§ΒΪ‘Ύ“ΜΕ®ΧθΦΰœ¬ΡޔꬻΤχΖΔ…ζ»Γ¥ζΖ¥”ΠΘ§Τδ“Μ¬»»Γ¥ζΈο÷Μ”–“Μ÷÷Θ§‘ρΧΰAΒΡΫαΙΙΦρ ΫΈΣ__________________ΓΘ
Θ®4Θ©»τΧΰAΡή ΙδεΥ°Ά …ΪΘ§‘Ύ¥ΏΜ·ΦΝΉς”Οœ¬Θ§”κH2Φ”≥…Θ§ΤδΦ”≥…≤ζΈοΨ≠≤βΕ®Ζ÷Ή”÷–Κ§”–4ΗωΦΉΜυΘ§ΧΰAΩ…Ρή”–ΒΡΫαΙΙΦρΦρ Ϋ__________________________________________ΓΘ
Θ®11Ζ÷Θ©Δ≈C6H12 Θ®1Ζ÷Θ© ΔΤ42 Θ®1Ζ÷Θ© 100.8 Θ®1Ζ÷Θ©
Δ« Θ®1Ζ÷Θ© Δ»(CH3)3CCH=CH2ΓΔCH2=C(CH3)CH(CH3)2ΓΔ(CH3)2C=C(CH3)2Θ®ΟΩΗω2Ζ÷Θ§Ι≤
Θ®1Ζ÷Θ© Δ»(CH3)3CCH=CH2ΓΔCH2=C(CH3)CH(CH3)2ΓΔ(CH3)2C=C(CH3)2Θ®ΟΩΗω2Ζ÷Θ§Ι≤
Δ«
 Θ®1Ζ÷Θ© Δ»(CH3)3CCH=CH2ΓΔCH2=C(CH3)CH(CH3)2ΓΔ(CH3)2C=C(CH3)2Θ®ΟΩΗω2Ζ÷Θ§Ι≤
Θ®1Ζ÷Θ© Δ»(CH3)3CCH=CH2ΓΔCH2=C(CH3)CH(CH3)2ΓΔ(CH3)2C=C(CH3)2Θ®ΟΩΗω2Ζ÷Θ§Ι≤‘ΧβΖ÷ΈωΘΚΘ®1Θ©ΧΰA 0.2 mol ‘Ύ―θΤχ÷–Άξ»Ϊ»Φ…’ΚσΘ§…ζ≥…CO2ΚΆH2OΗς1.2 molΘ§‘ρAΖ÷Ή”÷–ΧΦ«β‘≠Ή”ΒΡΗω ΐΖ÷±π «6ΗωΚΆ12ΗωΘ§‘ρΜ·―ß Ϋ”ΠΗΟ «C6H12ΓΘ
Θ®2Θ©ΧΰAΆξ»Ϊ»Φ…’ΚσΘ§…ζ≥…CO2ΚΆH2OΗς3 molΘ§‘ρΗυΨίAΒΡΜ·―ß ΫΩ…÷ΣΘ§≤ΈΦ”Ζ¥”ΠΒΡAΒΡΈο÷ ΒΡΝΩ «0.5molΘ§‘ρAΒΡ÷ ΝΩ «0.5molΓΝ84g/molΘΫ42gΓΘ
Θ®3Θ©ΧΰA≤ΜΡή ΙδεΥ°Ά …ΪΘ§ΥΒΟςΖ÷Ή”÷–ΟΜ”–ΧΦΧΦΥΪΦϋΓΘΒΪ‘Ύ“ΜΕ®ΧθΦΰœ¬ΡޔꬻΤχΖΔ…ζ»Γ¥ζΖ¥”ΠΘ§Τδ“Μ¬»»Γ¥ζΈο÷Μ”–“Μ÷÷Θ§’βΥΒΟςΖ÷Ή”ΒΡ«β‘≠Ή”÷Μ”–“ΜάύΘ§Υυ“‘A «ΜΖΦΚΆιΘ§ΫαΙΙΦρ Ϋ «
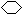 ΓΘ
ΓΘΘ®4Θ©»τΧΰAΡή ΙδεΥ°Ά …ΪΘ§ΥΒΟςΖ÷Ή” «Κ§”–ΧΦΧΦΥΪΦϋΒΡΓΘ‘Ύ¥ΏΜ·ΦΝΉς”Οœ¬Θ§”κH2Φ”≥…Θ§ΤδΦ”≥…≤ζΈοΨ≠≤βΕ®Ζ÷Ή”÷–Κ§”–4ΗωΦΉΜυΘ§“ρ¥ΥΧΰAΩ…Ρή”–ΒΡΫαΙΙΦρΦρ Ϋ(CH3)3CCH=CH2ΓΔCH2=C(CH3)CH(CH3)2ΓΔ(CH3)2C=C(CH3)2ΓΘ
ΒψΤάΘΚΗΟΧβ «ΗΏΩΦ÷–ΒΡ≥ΘΦϊΧβ–ΆΘ§ τ”Ύ÷–Β»Ρ―Ε»ΒΡ ‘ΧβΓΘ ‘ΧβΡ―“Ή ÷–Θ§ΉέΚœ–‘«ΩΘ§≤ύ÷ΊΕ‘―ß…ζΡήΝΠΒΡ≈ύ―χΚΆΫβΧβΖΫΖ®ΒΡ÷ΗΒΦ”κ―ΒΝΖΘ§”–÷ζ”Ύ≈ύ―χ―ß…ζΒΡ¬ΏΦ≠ΆΤάμΡήΝΠΚΆΖΔ…ΔΥΦΈ§ΡήΝΠΓΘΗΟΧβΒΡΙΊΦϋ «Ος»ΖΗς÷÷ΙΌΡήΆ≈ΫαΙΙΚΆ–‘÷ Θ§»ΜΚσΫαΚœΧβ“βΨΏΧεΈ ΧβΓΔΨΏΧεΖ÷ΈωΦ¥Ω…Θ§”–÷ζ”Ύ≈ύ―χ―ß…ζΒΡΡφœρΥΦΈ§ΡήΝΠΚΆ”Π ‘ΡήΝΠΓΘ

ΝΖœΑ≤αœΒΝ–¥πΑΗ
 ΩΣ–ΡΆήΉ¥‘Σ≤β ‘ΨμœΒΝ–¥πΑΗ
ΩΣ–ΡΆήΉ¥‘Σ≤β ‘ΨμœΒΝ–¥πΑΗ
œύΙΊΧβΡΩ
 Θ®Μ·―ß ΫΈΣC8H10OΘ©”–Εύ÷÷Ά§Ζ÷“λΙΙΧεΘ§Τδ÷– τ”ΎΖΦœψ¥ΦΒΡΆ§Ζ÷“λΙΙΧε“ΜΙ≤”–Θ® Θ©ΓΓ
Θ®Μ·―ß ΫΈΣC8H10OΘ©”–Εύ÷÷Ά§Ζ÷“λΙΙΧεΘ§Τδ÷– τ”ΎΖΦœψ¥ΦΒΡΆ§Ζ÷“λΙΙΧε“ΜΙ≤”–Θ® Θ©ΓΓ