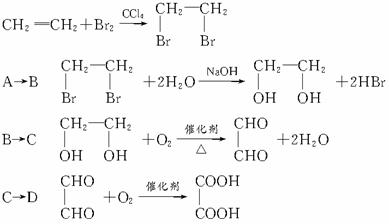
ΧβΡΩΡΎ»ί
”…““œ©ΚΆΤδΥϊΈόΜζ‘≠ΝœΚœ≥…ΜΖΉ¥Μ·ΚœΈοEΘ§ΤδΚœ≥…Ιΐ≥Χ»γœ¬ΘΚΘ®Υ°ΦΑΤδΥϊΈόΜζ≤ζΈοΨυ“― Γ¬‘Θ©
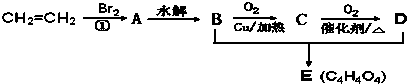
‘Ά®ΙΐΖ÷ΈωΜΊ¥πœ¬Ν–Έ ΧβΘΚ
Θ®1Θ©–¥≥ωCΒΡΫαΙΙΦρ Ϋ
Θ®2Θ©Έο÷ X”κAΜΞΈΣΆ§Ζ÷“λΙΙΧεΘ§‘ρXΒΡΫαΙΙΦρ ΫΈΣ
Θ®3Θ©÷Η≥ω…œΆΦ±δΜ·Ιΐ≥Χ÷–ΔΌΒΡ”–ΜζΖ¥”Πάύ–Ά
Θ®4Θ©DΈο÷ ÷–ΙΌΡήΆ≈ΒΡΟϊ≥Τ
Θ®5Θ©–¥≥ωBΚΆD…ζ≥…EΒΡΜ·―ßΖ¥”ΠΖΫ≥Χ Ϋ
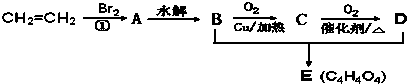
‘Ά®ΙΐΖ÷ΈωΜΊ¥πœ¬Ν–Έ ΧβΘΚ
Θ®1Θ©–¥≥ωCΒΡΫαΙΙΦρ Ϋ
OHC-CHO
OHC-CHO
ΘΜΘ®2Θ©Έο÷ X”κAΜΞΈΣΆ§Ζ÷“λΙΙΧεΘ§‘ρXΒΡΫαΙΙΦρ ΫΈΣ
CH3-CHBr2
CH3-CHBr2
ΘΜΘ®3Θ©÷Η≥ω…œΆΦ±δΜ·Ιΐ≥Χ÷–ΔΌΒΡ”–ΜζΖ¥”Πάύ–Ά
Φ”≥…
Φ”≥…
ΘΜΘ®4Θ©DΈο÷ ÷–ΙΌΡήΆ≈ΒΡΟϊ≥Τ
τ»Μυ
τ»Μυ
ΘΜΘ®5Θ©–¥≥ωBΚΆD…ζ≥…EΒΡΜ·―ßΖ¥”ΠΖΫ≥Χ Ϋ
CH2OHCH2OH+HCCOC-COOH
 +2H2O
+2H2O
| ≈®H2SO4 |
| Γς |
 +2H2O
+2H2OCH2OHCH2OH+HCCOC-COOH
 +2H2O
+2H2O
Θ®ΉΔΟςΖ¥”ΠΧθΦΰΘ©Θ°| ≈®H2SO4 |
| Γς |
 +2H2O
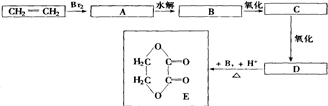
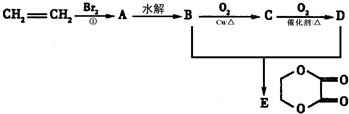
+2H2OΖ÷ΈωΘΚCH2=CH2ΚΆδεΖΔ…ζΦ”≥…Ζ¥”ΠΘ§…ζ≥…AΈΣCH2BrCH2BrΘ§Υ°Ϋβ…ζ≥…BΈΣCH2OHCH2OHΘ§―θΜ·≤ζΈοCΈΣOHC-CHOΘ§ΫχΕχ±Μ―θΜ·ΈΣDΘ§ΈΣHCCOC-COOHΘ§ΫαΚœ”–ΜζΈοΒΡΫαΙΙΚΆ–‘÷ Ω…Ϋβ¥πΗΟΧβΘ°
Ϋβ¥πΘΚΫβΘΚCH2=CH2ΚΆδεΖΔ…ζΦ”≥…Ζ¥”ΠΘ§…ζ≥…AΈΣCH2BrCH2BrΘ§Υ°Ϋβ…ζ≥…BΈΣCH2OHCH2OHΘ§―θΜ·≤ζΈοCΈΣOHC-CHOΘ§ΫχΕχ±Μ―θΜ·ΈΣDΘ§ΈΣHCCOC-COOHΘ§
Θ®1Θ©”…“‘…œΖ÷ΈωΩ…÷ΣCΈΣOHC-CHOΘ§Ι ¥πΑΗΈΣΘΚOHC-CHOΘΜ
Θ®2Θ©AΈΣCH2BrCH2BrΘ§Ε‘”ΠΒΡΆ§Ζ÷“λΙΙΧεΈΣCH3-CHBr2Θ§Ι ¥πΑΗΈΣΘΚCH3-CHBr2ΘΜ
Θ®3Θ©Ζ¥”ΠΔΌΈΣΦ”≥…Ζ¥”ΠC=C…ζ≥…C-CΘ§Ι ¥πΑΗΈΣΘΚΦ”≥…ΘΜ
Θ®4Θ©DΈΣHCCOC-COOHΘ§Κ§”–ΒΡΙΌΡήΆ≈ΈΣτ»ΜυΘ§Ι ¥πΑΗΈΣΘΚτ»ΜυΘΜ
Θ®5Θ©BΈΣCH2OHCH2OHΘ§DΈΣHCCOC-COOHΘ§Εΰ’ΏΖΔ…ζθΞΜ·Ζ¥”Π…ζ≥…ΜΖθΞΘ§Ζ¥”ΠΒΡΖΫ≥Χ ΫΈΣCH2OHCH2OH+HCCOC-COOH
 +2H2Oȧ
+2H2Oȧ
Ι ¥πΑΗΈΣΘΚCH2OHCH2OH+HCCOC-COOH
 +2H2OΘ°
+2H2OΘ°
Θ®1Θ©”…“‘…œΖ÷ΈωΩ…÷ΣCΈΣOHC-CHOΘ§Ι ¥πΑΗΈΣΘΚOHC-CHOΘΜ
Θ®2Θ©AΈΣCH2BrCH2BrΘ§Ε‘”ΠΒΡΆ§Ζ÷“λΙΙΧεΈΣCH3-CHBr2Θ§Ι ¥πΑΗΈΣΘΚCH3-CHBr2ΘΜ
Θ®3Θ©Ζ¥”ΠΔΌΈΣΦ”≥…Ζ¥”ΠC=C…ζ≥…C-CΘ§Ι ¥πΑΗΈΣΘΚΦ”≥…ΘΜ
Θ®4Θ©DΈΣHCCOC-COOHΘ§Κ§”–ΒΡΙΌΡήΆ≈ΈΣτ»ΜυΘ§Ι ¥πΑΗΈΣΘΚτ»ΜυΘΜ
Θ®5Θ©BΈΣCH2OHCH2OHΘ§DΈΣHCCOC-COOHΘ§Εΰ’ΏΖΔ…ζθΞΜ·Ζ¥”Π…ζ≥…ΜΖθΞΘ§Ζ¥”ΠΒΡΖΫ≥Χ ΫΈΣCH2OHCH2OH+HCCOC-COOH
| ≈®H2SO4 |
| Γς |
 +2H2Oȧ
+2H2OΘ§Ι ¥πΑΗΈΣΘΚCH2OHCH2OH+HCCOC-COOH
| ≈®H2SO4 |
| Γς |
 +2H2OΘ°
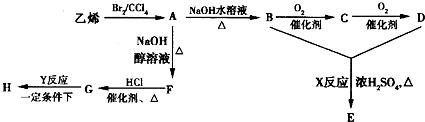
+2H2OΘ°ΒψΤάΘΚ±ΨΧβΩΦ≤ι”–ΜζΈοΒΡΚœ≥…Θ§ΧβΡΩΡ―Ε»≤Μ¥σΘ§ΉΔ“βΑ―Έ’≥ΘΦϊ”–ΜζΈοΒΡ–‘÷ “‘ΦΑΖ¥”ΠΧθΦΰΚΆΖ¥”Πάύ–ΆΘ§ΈΣ÷–―ßΫΉΕΈΩΦ ‘»»ΒψΈ ΧβΘ§‘Ύ―ßœΑ÷–ΉΔ“βΜΐάέΘ°

ΝΖœΑ≤αœΒΝ–¥πΑΗ
œύΙΊΧβΡΩ

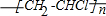 Θ§
Θ§ +2NaOH
+2NaOH