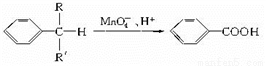��Ŀ����
����Se������һ�ַǽ���Ԫ�ء����������������ϡ��������ҵ����������������Ӫ��Ԫ�غ�ֲ�������Ӫ��Ԫ�صȡ���ش��������⣺
(1) Seԭ�ӵĺ�������Ų�ʽΪ____________��
(2) �Ƚ�As�ĵ�һ�����ܱ�Se______�����С������
(3) SeO42-��Se-O�ļ��DZ�SeO3�ļ���____(���С��)ԭ����______________
(4) H2SeO4��H2SeO3ˮ��Һ������___________��
(5) �������Ǻ�ɫ���ɫ��ĩ������ɫ��������������������ľ���Ϊ���������ṹ��ԭ������Ϊ�������������ֲ������������ϣ�ͬһ������ԭ�����ú�ǿ��������֮��ԭ�����ý�������������״ͼ�������ṹͼ�;�������ͼ���¡�
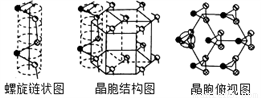
��������Seԭ�ӵ��ӻ���ʽΪ__________����֪���������ı߳�Ϊacm����Ϊb cm�������ӵ�����ΪNAmol-1����þ����к��е�Seԭ�Ӹ���Ϊ_________���þ�����ܶ�Ϊ___________g/cm3 (�ú�NA��a��b��ʽ�ӱ�ʾ)��
���и��������У����ܰ�ͼʾ����������ʾһ����ɣ���ϵ�ת������
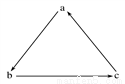
ѡ�� | a | b | c |
A | Fe | FeCl3 | FeCl2 |
B | H2SO4 | SO2 | SO3 |
C | Si | SiO2 | H2SiO3 |
D | HNO3 | NO | NO2 |
A. A B. B C. C D. D
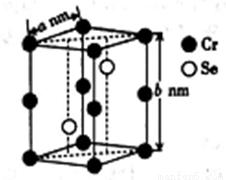
�������ʮ�����ĵز��У�����Ԫ�ش�ǧ��֮������ͭ��61����������(Ti)����Ϊ21���ͽ������䵥�ʺͻ�������й㷺��Ӧ�ü�ֵ��������(Ti3N4)Ϊ���ɫ���壬���ھ�����������ķ½�Ч����Խ��Խ��س�Ϊ�ƽ�Ĵ���Ʒ����TiCl4Ϊԭ�ϣ�����һϵ,�з�Ӧ�����Ƶ�Ti3N4������TiO2(����ͼ)��
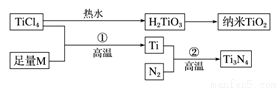
ͼ�е�M�Ƕ����ڽ���Ԫ�أ�M�IJ��ֵ��������±�:
I1 | I2 | I3 | I4 | I5 | |
������/(kJ��mol-1) | 738 | 1451 | 7733 | 10 540 | 13 630 |
��ش��������⣺
��TiO2Ϊ���Ӿ��壬��֪�����������ӵ���λ��Ϊ6�������ӵ���λ��Ϊ3���������ӵĵ����Ų�ʽΪ___________
�ƽ���Ti�����M�ľ���ԭ�Ӷѻ�ģʽ��ͬ����ѻ�ģ��Ϊ_________����д�ѻ�ģ������)��������ԭ���ڶ�άƽ�������λ��Ϊ_____________
��������TiCl4Ϊ��ɫҺ�壬�е�Ϊ136.4�棬�ɴ˿�֪�侧������Ϊ__________�����ɸþ�������ӵĿռ乹��Ϊ_____________
(4)��һ�ֵ����Ѿ���ľ�����NaCl�������ƣ�����ͼ��ʾ����֪�õ����ѵ��ܶ�Ϊp g��cm-3����þ�����N��Ti֮����������Ϊ_____ pm����NAΪ�����ӵ³�������ֵ��ֻ����ʽ�����þ�������Tiԭ�Ӿ�������������Tiԭ����___________����
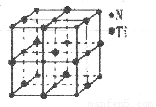
(5)��ѧ��ͨ��X����̽��KC1��MgO��CaO��TiN�ľ�����NaCl�ľ���ṹ���ơ���֪�������Ӿ���ľ������������£�
���Ӿ��� | NaCl | KC1 | CaO |
������/(kJ��mol-1) | 786 | 715 | 3401 |
KC1��CaO��TiN�������Ӿ���Ӳ���ɵ͵��õ�˳��Ϊ_____________


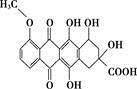 ���йظ����ʵ�˵������ȷ����( ��
���йظ����ʵ�˵������ȷ����( ��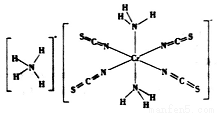
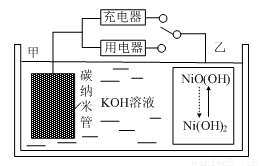
 2Ni(OH)2
2Ni(OH)2