��Ŀ����
(1)����ʱ�������Ƶ��ܶȻ�Ksp��4.7��10��6������ʱ��9mL 0.02mol��L��1���Ȼ�����Һ��1mL pH��13������������Һ��Ϻ�(��Һ�����ֱ�ӼӺ�)����Һ��
������������(�����)��
(2)2 .24��(��״̬)����ͨ��250mLŨ��Ϊ0.1mol��L��1��������Һ�У���ַ�Ӧ����Һ�и�����Ũ���ɴ�С��˳��Ϊ������
.24��(��״̬)����ͨ��250mLŨ��Ϊ0.1mol��L��1��������Һ�У���ַ�Ӧ����Һ�и�����Ũ���ɴ�С��˳��Ϊ������
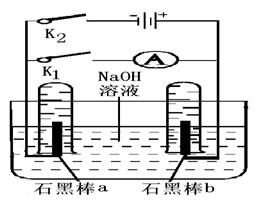
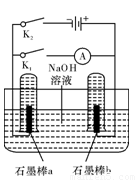
(3)����ͼ�����Ӧʵ�顣
�ٶϿ�K1���պ�K2��ֱͨ����Դ��д�������ĵ缫��Ӧʽ�� ��
�ڵ��һ��ʱ�����ʯī�����������Χʱ���ж�K2�պ�K1�����ֵ� ·���е���ͨ����д��a���ĵ缫��Ӧʽ ����
·���е���ͨ����д��a���ĵ缫��Ӧʽ ����
(1)���ޡ�
(2)��c(NH)>c(SO)>c(OH��)>c(H��)����
(3)
�١�4OH����4e��===2H2O��O2������
�ڡ�2H2��4OH����4e��===4H2O����
����

��ϰ��ϵ�д�
�����Ŀ