��Ŀ����
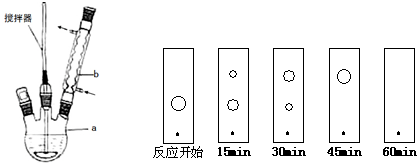
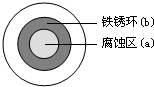 ��2011?�㽭����NaCl��Һ����һ�����������������ϣ�һ��ʱ�����Һ�θ��ǵ�Բ����������a���ѱ���ʴ���䰵����Һ��������ɫ�����b������ͼ��ʾ�����¸��������Ҫԭ����Һ��֮�����������ȱ�Ե�٣�����˵����ȷ���ǣ�������
��2011?�㽭����NaCl��Һ����һ�����������������ϣ�һ��ʱ�����Һ�θ��ǵ�Բ����������a���ѱ���ʴ���䰵����Һ��������ɫ�����b������ͼ��ʾ�����¸��������Ҫԭ����Һ��֮�����������ȱ�Ե�٣�����˵����ȷ���ǣ�������������NaCl��Һ�ε�һ�����������������ϣ�һ��ʱ�����Һ�θ��ǵ�Բ����������a������ʴ�䰵��ʵ�����Ƿ�����������ʴ����ʱ�������缫��ӦΪ��Fe-2e-=Fe2+ ������������Ӧ�������缫��ӦΪ��O2+2H2O+4e-=4OH- ��������ԭ��Ӧ����Һ�����أ�����Fe2++2OH-=Fe��OH��2��4Fe��OH��2+O2+2H2O=4Fe��OH��3 �γ�����ɫ�����b����
����⣺A��������ԭ����У����������������������������Ĺ��ɣ�Cl-Ӧ��b����a��Ǩ�ƣ���A����
B��O2��Һ�����ط�Ӧ�������缫��ӦΪ��O2+2H2O+4e-=4OH- ��������ԭ��Ӧ������B��ȷ��
C��Һ���µ�Fe����������Ӧ������ʴ����C����
D��������Ƕ��һͭ��˿�������壬��ͭ���Ӵ����μ�NaCl��Һ������Fe�Ľ�����Ա�ͭǿ��Fe��Ϊ���������������ĵ缫��ӦΪ��Fe-2e-=Fe2+����D����
��ѡB��
B��O2��Һ�����ط�Ӧ�������缫��ӦΪ��O2+2H2O+4e-=4OH- ��������ԭ��Ӧ������B��ȷ��
C��Һ���µ�Fe����������Ӧ������ʴ����C����
D��������Ƕ��һͭ��˿�������壬��ͭ���Ӵ����μ�NaCl��Һ������Fe�Ľ�����Ա�ͭǿ��Fe��Ϊ���������������ĵ缫��ӦΪ��Fe-2e-=Fe2+����D����
��ѡB��
���������⿼��������ԭ��Ӧ���绯ѧ�ȷ���֪ʶ��ע����ԭ����У�����������������������������

��ϰ��ϵ�д�
 ����νӽ̲���ĩ���Ԥϰ�人������ϵ�д�
����νӽ̲���ĩ���Ԥϰ�人������ϵ�д� ������ҵ��ٳɳ����½������������ϵ�д�
������ҵ��ٳɳ����½������������ϵ�д�
�����Ŀ
 ��2011?�㽭��ij�о�С����ʵ����̽����������泥�NH2COONH4���ֽⷴӦƽ�ⳣ����ˮ�ⷴӦ���ʵIJⶨ��
��2011?�㽭��ij�о�С����ʵ����̽����������泥�NH2COONH4���ֽⷴӦƽ�ⳣ����ˮ�ⷴӦ���ʵIJⶨ��
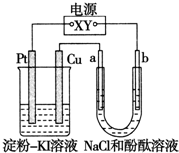 ��2011?�㽭��У����������CuI��һ�ֲ�����ˮ�İ�ɫ���壬�������ɷ�Ӧ��2Cu2++4I-�T2CuI��+I2���õ�����ͼ��ʾװ���У�a��b���Ƕ��Ե缫��ͨ��һ��ʱ����ڵ���-KI��Һ��������Χ����ɫ��������˵����ȷ���ǣ�������
��2011?�㽭��У����������CuI��һ�ֲ�����ˮ�İ�ɫ���壬�������ɷ�Ӧ��2Cu2++4I-�T2CuI��+I2���õ�����ͼ��ʾװ���У�a��b���Ƕ��Ե缫��ͨ��һ��ʱ����ڵ���-KI��Һ��������Χ����ɫ��������˵����ȷ���ǣ�������