��Ŀ����
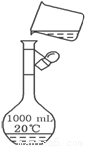
��14�֣�ʵ������Ҫ0.80 mol/L NaOH��Һ475 mL��0.40mol/L������Һ500 mL��������������Һ����������ش��������⣺
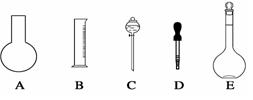
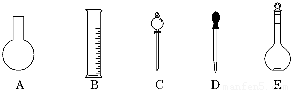
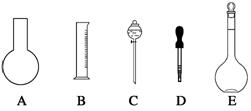
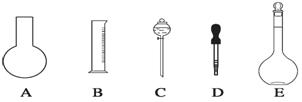
��1����ͼ��ʾ��������������Һ�϶�����Ҫ���� ������ţ�������������Һ�����õ��IJ��������� �����������ƣ���
��2�����в����У�����ƿ�����߱��Ĺ����� ������ţ���
A������һ�����ȷŨ�ȵı���Һ
B����ȡһ�������Һ��
C����������ƿ������µ����������Һ��
D��ȷϡ��ijһŨ�ȵ���Һ
E��������Һ
F�����������ܽ��������
��3�����ݼ�����������ƽ��ȡNaOH������Ϊ g����ʵ����������������ȷ��������ʱ�����ӿ̶��ߣ���������ҺŨ�� 0.8 mol/L������ڡ��������ڡ���С�ڡ�,��ͬ����������ʱ������������ˮ����������ƿ�⣬��������ҺŨ�� 0.8 mol/L��
��4�����ݼ����֪��������������Ϊ98%���ܶ�Ϊ1.84 g/cm3��Ũ��������Ϊ mL������������һλС���������ʵ������10 mL��15 mL��20 mL��50 mL��Ͳ��Ӧѡ�� mL��Ͳ��á�
��1��A��C��2�֣��ձ�����������2�֣���2��C��E��F��3�֣���3��16.0 ��2�֣����ڣ�1�֣����ڣ�1�֣���4��10.9��2�֣�15��1�֣�
����:

 ѧ���쳵�����ּ��������ҵ�½����������ϵ�д�
ѧ���쳵�����ּ��������ҵ�½����������ϵ�д� �����ѧСѧ�꼶�νӵ������㽭��ѧ������ϵ�д�
�����ѧСѧ�꼶�νӵ������㽭��ѧ������ϵ�д� Сѧ�����ҵ���ϴ�ѧ������ϵ�д�
Сѧ�����ҵ���ϴ�ѧ������ϵ�д� ���Ž�����ٰθ��νӹ㶫���������ϵ�д�
���Ž�����ٰθ��νӹ㶫���������ϵ�д���14�֣�ʵ������Ҫ0.80 mol/L NaOH��Һ475 mL��0.40 mol/L������Һ500 mL��������������Һ����������ش��������⣺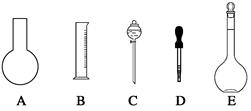
��1����ͼ��ʾ��������������Һ�϶�����Ҫ���� ������ţ�������������Һ�����õ��IJ��������� �����������ƣ���
��2�����в����У�����ƿ�����߱��Ĺ����� ������ţ���
| A������һ�����ȷŨ�ȵı���Һ |
| B����ȡһ�������Һ�� |
| C����������ƿ������µ����������Һ�� |
| D��ȷϡ��ijһŨ�ȵ���Һ |
F�����������ܽ��������
��3�����ݼ�����������ƽ��ȡNaOH������Ϊ g����ʵ����������������ȷ��������ʱ�����ӿ�
 ���ߣ���������ҺŨ�� 0.8 mol/L������ڡ��������ڡ���С�ڡ�,��ͬ����������ʱ������������ˮ����������ƿ�⣬��������ҺŨ�� 0.8 mol/L��
���ߣ���������ҺŨ�� 0.8 mol/L������ڡ��������ڡ���С�ڡ�,��ͬ����������ʱ������������ˮ����������ƿ�⣬��������ҺŨ�� 0.8 mol/L����4�����ݼ����֪��������������Ϊ98%���ܶ�Ϊ1.84 g/cm3��Ũ��������Ϊ mL������������һλС���������ʵ������10 mL��15 mL��20 mL��50 mL��Ͳ��Ӧѡ�� mL��Ͳ��á�