��Ŀ����
��1��37Cl�����ӽṹʾ��ͼ ��������ȵ���������SO2��SO3��������ԭ�Ӹ���֮
Ϊ ��
��2��20.6g NaR ����Na+0.2mol����NaR��Ħ������Ϊ ����R 8.0g ��NaR�����ʵ���Ϊ ��
��3���ڱ�״���£�4.8g����(CH4)��ռ�����Ϊ_________L�������״����________L����(H2S)������ͬ��Ŀ����ԭ�ӣ�
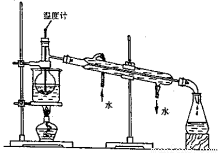
��4����100gŨ��Ϊ18 mol��L��1���ܶ�Ϊ��(g��cm�C3)��Ũ�����м���һ������ˮϡ�ͳ�9mol��L��1�����ᣬ�����ˮ����� 100mL����������������������������
��1��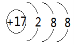 5: 6
5: 6
��2��103 g/mol����λ��д���÷֣��� 0.1 mol
��3��6.72�� 13.44
��4����
��������
�����������1��Cl?Ϊ18���ӣ������������Ϊ8��������ȵ���������SO2��SO3��������ԭ�Ӹ���֮Ϊm��64��2��m��80��3=5:6��
��2��Na+Ϊ0.2mol����NaRΪ0.2mol��M(NaR)=20.6g��0.2mol=103g/mol����NaR��Ħ�������ɵ�M(R)=80g/mol����n(NaR)=n(R)=8.0g��80g/mol=0.1mol��
��3����������Ħ��������м��㣬V(CH4)=4.8g��16g/mol��22.4L/mol=6.72L��4.8g��16g/mol��4=V(H2S)��22.4L/mol��2,�ɵ�V(H2S)=13.44L��
��4����18 mol��L-1H2SO4ϡ��Ϊ9mol��L-1��Ӧʹ��Һ�����Ϊԭ����2������Ϊ������Һ���ܶ�����Ũ�ȵļ�С����С��ϡ�ͺ��������Һ����С��ԭ��Һ��2�������Լ���ˮ�����С��100ml��
���㣺���⿼�黯ѧ�����ѧ���㡣

 Сѧ�̲�ȫ��ϵ�д�
Сѧ�̲�ȫ��ϵ�д� Сѧ��ѧ������ѿڶ���ϵ�д�
Сѧ��ѧ������ѿڶ���ϵ�д� ������Ӧ�������������ϵ�д�
������Ӧ�������������ϵ�д� �㽭֮�ǿ�ʱ�Ż���ҵϵ�д�
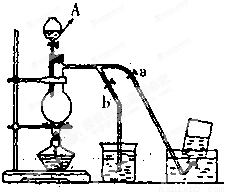
�㽭֮�ǿ�ʱ�Ż���ҵϵ�д����������ѧУ��ѧʵ����װ�ޣ�ҩƷ��ת�Ƶ��˰�ȫ�ij������ܣ�������Ϊ������Ա��������м�ƿ��������û�м�ʱת�ƣ���ѧ��ŷ��֣�������Ҫ�ⶨ���������Ƿ��б��ʣ������ʲô������ij��ȤС�������������������ô���ģ������£���ȡ��ͬ����������Ʒ����ˮ���������к���pH=7����Ӧ������û�й۲쵽���ݣ�Ȼ����Һ���ɵ��Ȼ��ƾ��壬���ɹ����в�Ʒ����ʧ��
| ������������(g) | �Ȼ�������(g) |
�� | 2.40 | 3.51 |
�� | 2.32 | 2.34 |
�� | 3.48 | 3.51 |
����ʵ���٢ڢ����ݿɿ���ͨ�����㣬�����ͱȽ��ϱ�3�����ݣ��������ۡ�