��Ŀ����
�������ӷ���ʽ��д����ȷ���� �� ��
| A���������ۼ���ϡ�����У�3Fe��8H����2NO3��=3Fe2����2NO����4H2O |
| B��Ca��HCO3��2��Һ�����Ca��OH��2��Һ��Ӧ��Ca2����HCO3����OH��=CaCO3����H2O |
| C��NH4HCO3��Һ����Ba��OH��2��Һ����:2HCO3һ��2OH����Ba2��=BaCO3����CO32����2H2O |
| D��Na2S��Һ��ͨ������H2S��S2һ��H2S=2HS�� |
AC
��

��ϰ��ϵ�д�
�����Ŀ
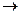 H2O
H2O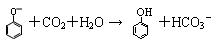
 NH3����2H2O
NH3����2H2O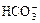 ��OH-��CaCO3¯��H2O
��OH-��CaCO3¯��H2O H2��+ Cu2+
H2��+ Cu2+ CaCO3����H2O
CaCO3����H2O Cu2+��SO2����2H2O
Cu2+��SO2����2H2O