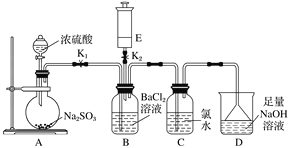
��Ŀ����
����Ŀ������ȷ��ʾ���з�Ӧ�����ӷ���ʽ��
A. �ù���ʯ�������չ�ҵβ���е�SO2: Ca2++2OH-+SO2��CaSO3 ��+ H2O
B. ������KMnO4��Һ��H2O2��Ӧ��֤��H2O2���л�ԭ�ԣ�2MnO4��+6H++5H2O2��2Mn2++5O2 ��+8H2O
C. ��ͭ���缫���NaCl��Һ:2C1��+2H2O![]() H2��+Cl2��+2OH-
H2��+Cl2��+2OH-
D. �� Fe2O3���뵽 HI ��Һ�У�Fe2O3+ 6H+��2Fe3+ +3H2O
���𰸡�B
�����������������A��ʯ�����е���������ֻ�������ܽ⣬�û�ѧʽ��ʾ����A����B������KMnO4���������ԣ���H2O2��Ӧ����������ԭ��Ӧ���ų���������B��ȷ��C��ͭ�ǻ��Ե缫��������ʱ��ͭҪʧȥ���Ӷ��ܽ⣬��C����D�����ɵ�Fe3+���������ԣ��ܹ����������ӣ���D����ѡB��

��ϰ��ϵ�д�
�����Ŀ