��Ŀ����
(18��)MnO2��Zn������ɵ�ص���Ҫԭ�ϣ���ҵ�������̿�(��Ҫ�ɷ�MnO2)����п��(��Ҫ�ɷ�ZnS)��������MnO2��Zn�Ĺ�����������ͼ��ʾ��
��1������I�õ������β��ʵ��������ձ�����������_______ _____(����������)��
��2��ϡ�������ʱ��Ӧ�����ӷ���ʽΪ_______________________________________���÷�Ӧ��������19.2g����A����ת��____________mo1���ӡ�����ʱ��Ӧ���ʽ��������д�ʩ������߽���ʱ��Ӧ���ʵ���________(�����)��
a������ʯ����
b����߽����¶�
c���ʵ���������Ũ��
d���ı����̿�����п��ı���
��3������������Һ�ɵõ�����̼���̣�Ȼ���ڿ���������̼�����Ʊ�MnO2����֪��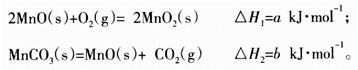
д��̼�����ڿ�������������MnO2���Ȼ�ѧ����ʽ_________________________��
��4���ö��Ե缫��������ữ����������Һ�Ʊ�MnO2��װ������ͼ��ʾ��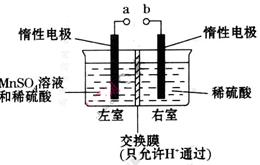
��aӦ��ֱ����Դ��_________(���������)����������
�ڵ������������ӵ�������______________��_____________����ת�Ƶĵ�����Ϊ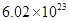 ����������Һ��
����������Һ��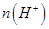 �ı仯��Ϊ________________��
�ı仯��Ϊ________________��
��1��©��
��2��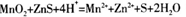 ��1.2��d
��1.2��d
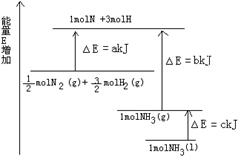
��3��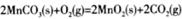 ��H=��a+2b��kJ/mol��
��H=��a+2b��kJ/mol��
��4�������ڲ���������Ӧ��ͨ������Ĥ�����ƶ��γɵ�����1mol
���������������1������IΪ���ˣ��õ������β��ʵ��������ձ�����������©������2��ϡ�������ʱ��ӦΪMnO2��ZnS��ΪMn2+��Zn2+��ͬʱӦ����S���ʣ��ʷ���ʽΪ��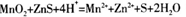 ��19.2g����S�����ʵ���Ϊ0.6mol���ɷ�Ӧ����ʽ��֪����1molSת��2mole-,������0.6molSת��1.2mole-���ĸ�ѡ���У�����ʯ���顢��߽����¶ȡ��ʵ���������Ũ�Ⱦ�����߷�Ӧ���ʣ��ʴ�Ϊd����3���ɸ�˹���ɽ���ӦI+II��2�ɵ�
��19.2g����S�����ʵ���Ϊ0.6mol���ɷ�Ӧ����ʽ��֪����1molSת��2mole-,������0.6molSת��1.2mole-���ĸ�ѡ���У�����ʯ���顢��߽����¶ȡ��ʵ���������Ũ�Ⱦ�����߷�Ӧ���ʣ��ʴ�Ϊd����3���ɸ�˹���ɽ���ӦI+II��2�ɵ�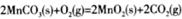 ��H=��a+2b��kJ/mol����4���ٸ���װ��ͼ��֪���ҷ���MnSO4��MnO2�ķ�Ӧ��������������Ӧ����aӦ��ֱ����Դ�������������ڵ�����������ΪH+�õ��ӷ�����ԭ��Ӧ���ֽ���Ĥֻ����H+ͨ������H+������Ϊ����������Ӧ��ͨ������Ĥ�����ƶ��γɵ�������ת�Ƶĵ�����Ϊ
��H=��a+2b��kJ/mol����4���ٸ���װ��ͼ��֪���ҷ���MnSO4��MnO2�ķ�Ӧ��������������Ӧ����aӦ��ֱ����Դ�������������ڵ�����������ΪH+�õ��ӷ�����ԭ��Ӧ���ֽ���Ĥֻ����H+ͨ������H+������Ϊ����������Ӧ��ͨ������Ĥ�����ƶ��γɵ�������ת�Ƶĵ�����Ϊ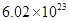 ����1mol�����ݵ���غ����������ʵ����仯Ϊ1mol��
����1mol�����ݵ���غ����������ʵ����仯Ϊ1mol��
���㣺���黯ѧʵ��������������ӷ���ʽ����д��������ԭ��Ӧ����ѧ��Ӧ����Ӱ�����ء���˹���ɡ��绯ѧ��֪ʶ��

 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д���֪298 K,101 kPaʱ��2SO2(g)��O2(g)  2SO3(g)����H����197 kJ��mol��1������ͬ�¶Ⱥ�ѹǿ�£����ܱ�������ͨ��2 mol SO2��1 mol O2���ﵽƽ��ʱ���ų�����ΪQ1������һ�������ͬ��������ͨ��1 mol SO2,0.5 mol O2��1 mol SO3���ﵽƽ��ʱ�ų�����ΪQ2�������й�ϵ��ȷ����(����)
2SO3(g)����H����197 kJ��mol��1������ͬ�¶Ⱥ�ѹǿ�£����ܱ�������ͨ��2 mol SO2��1 mol O2���ﵽƽ��ʱ���ų�����ΪQ1������һ�������ͬ��������ͨ��1 mol SO2,0.5 mol O2��1 mol SO3���ﵽƽ��ʱ�ų�����ΪQ2�������й�ϵ��ȷ����(����)
| A��Q2��Q1��197 kJ��mol��1 | B��Q2��Q1��197 kJ��mol��1 |
| C��Q1��Q2��197 kJ��mol��1 | D��Q2��Q1��197 kJ��mol��1 |
��7�֣����о���ѧ��Ӧ�е������仯ʱ������ͨ���������ʵ�飺
��һ��С�ձ������20 g����ĥ�ɷ�ĩ��Ba(OH)2��8H2O����С�ձ����������ѵ���3��4��ˮ�IJ���Ƭ�ϣ�Ȼ�����ձ��м���Լ10 g NH4Cl���壬����ʵ�鲽�裬��д�±������ش����⡣
| ʵ�鲽�� | ʵ�������� |
| �������ϣ��������ٽ��� | �д̼�����ζ��ʹʪ�����ɫʯ����ֽ������___��__���� |
| �������ձ��²� | �о��ձ�����˵���˷�Ӧ��_ �� ��Ӧ |
| ���������ձ� | �ձ�����Ĵ��м���ˮ�IJ���Ƭճ�����ձ��ײ� |
| ��ճ�в���Ƭ���ձ�����ʢ����ˮ���ձ��� | ����Ƭ���������ձ��ײ� |
| ��Ӧ�����߶������Ƭ�۲췴Ӧ�� | �����ɺ�״��֤����_��___���� |
�� �� ��
��2��ʵ����Ҫ�����ò�����Ѹ�ٽ����ԭ���ǣ� __________________����2�֣�
��3��������ʵ������У�Ϊʲô�ý���ϡ�����ʪ�����ڶ�����ϰ��ϣ���2�֣�
___________________________________________________________��
��14�֣������ѣ�CH3OCH3���ͼ״���CH3OH�����Ǹ�Ч�����Դ����ҵ������ú���������ˮú�����ϳɼ״��Ͷ����ѡ��ش��������⣺
��1���Ʊ����������һ����Ӧ��Al2O3���״���ˮ�ϳɣ���Ӧ����ʽΪ ��
��2����֪��CO(g)+2H2(g)=CH3OH (g) ��H= ��90.1kJ��mol-1 CO(g)��ȼ������282.8 kJ��mol-1��H2��ȼ������285.8 kJ��mol-1д����ʾCH3OH (g) ȼ���ȵ��Ȼ�ѧ��Ӧ����ʽ ��
��3��������ֱ��ȼ�ϵ�رȼ״�ֱ��ȼ�ϵ�ظ���Ч���������Ķ����Ѻͼ״���ȫ�ŵ�ת�Ƶ��ӵ����ʵ���֮���� ���ö�����ֱ��ȼ�ϵ�ص����������ʳ��ˮ��������9.2g������ʱ������������������������Ϊ L��������£�
��4���ںϳ��а���ˮú��������Ӧ��CO(g)+H2O(g)  CO2(g)+H2(g)�������ʵ�����CO(g)��H2O(g)�����ܱ������з�Ӧ��ƽ��ʱ��ý�����±���
CO2(g)+H2(g)�������ʵ�����CO(g)��H2O(g)�����ܱ������з�Ӧ��ƽ��ʱ��ý�����±���
| �¶� | 260�� | 280�� | 295�� | 310�� |
| COת���� | 89% | 80% | 75% | 60% |
����ʽ����280��ʱƽ�ⳣ�� ��
����ƽ����ϵ�У����H2��ѹǿռ��ѹ��30%��Ҫʹ��ϵ��COת���ʴﵽ70%��Ӧ��ʹ�¶� ������ߡ��������͡��������䡱��

 2SO3��g��
2SO3��g�� 2SO3��g�� ��H�� ��
2SO3��g�� ��H�� ��
 CO(g)��3H2(g)���÷�Ӧ��H����206 kJ��mol��1��
CO(g)��3H2(g)���÷�Ӧ��H����206 kJ��mol��1��