题目内容
已知氮的氧化物跟NaOH溶液发生的化学反应如下:
3NO2+2NaOH===2NaNO3+NO↑+H2O NO2+NO+2NaOH===2NaNO2+H2O
现有m mol NO2和n mol NO组成的混合气体,要用NaOH溶液使其完全吸收,无气体剩余。现有浓度为a mol/L的NaOH溶液,则需此NaOH溶液的体积为
3NO2+2NaOH===2NaNO3+NO↑+H2O NO2+NO+2NaOH===2NaNO2+H2O
现有m mol NO2和n mol NO组成的混合气体,要用NaOH溶液使其完全吸收,无气体剩余。现有浓度为a mol/L的NaOH溶液,则需此NaOH溶液的体积为
[ ]
A. L
L
B. L
L
C. L
L
D. L
L
 L
L B.
 L
L C.
 L
L D.
 L
L D

练习册系列答案
 天天向上口算本系列答案
天天向上口算本系列答案
相关题目
 L
L L
L L
L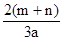 L
L L B.
L B. L C.
L C. L D.
L D. L
L