��Ŀ����
����P(s)��Cl2(g)������Ӧ����PCl3(g)��PCl5(g)����Ӧ���̺�������ϵ��ͼ��ʾ��ͼ�еġ�H��ʾ����1mol��������ݣ���
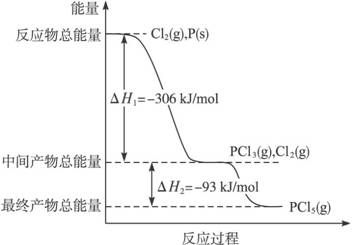
������ͼ�ش��������⣺
��1��P��Cl2��Ӧ����PCl3���Ȼ�ѧ����ʽ��
��������������������������������������������������������������������������
��2��PCl5�ֽ��PCl3��Cl2���Ȼ�ѧ����ʽ��
��������������������������������������������������������������������������
�����ֽⷴӦ��һ�����淴Ӧ���¶�T1ʱ�����ܱ������м���0.80mol PCl5,��Ӧ��ƽ��ʱPCl5��ʣ0.60 mol����ֽ�����1������������������Ӧ�¶���T1���ߵ�T2��ƽ��ʱPCl5�ķֽ���Ϊ��2����2 ��1(����ڡ�����С�ڡ����ڡ�)��
��3����ҵ���Ʊ�PCl5ͨ�����������У��Ƚ�P��Cl2��Ӧ�����м����PCl3��Ȼ���£��ٺ�Cl2��Ӧ����PCl5��ԭ����________________________��������������������������������������������
��4��P��Cl2��������Ӧ����1mol PCl5�ġ�H3=____________��P��Cl2һ����Ӧ����1molPCl5�ġ�H4__________��H3(����ڡ�����С�ڡ����ڡ�)��
��5��PCl5������ˮ��ַ�Ӧ���������������ᣬ�仯ѧ����ʽ��__________________________________��
��1��![]() Cl2 (g)�� P(s)
Cl2 (g)�� P(s)![]() PCl3 (g) ��H����306 kJ/ lmol������������������������
PCl3 (g) ��H����306 kJ/ lmol������������������������
��2�� PCl5(g)![]() PCl3(g)��Cl2(g) ��H��93kJ/ mol����������������������������
PCl3(g)��Cl2(g) ��H��93kJ/ mol����������������������������
25�������������ڡ�������������������������������������������������
��3��������Ӧ��Ϊ���ȷ�Ӧ��������������߲��ʣ���ֹ����ֽ⡡����������
��4����399kJ/ mol������ ������
��5��PCl5��4H2O��H3PO4��5HCl
��������1����ͼ��֪P��Cl2��Ӧ����PCl3���Ȼ�ѧ����ʽ��P��s)+![]() Cl2(g)
Cl2(g)![]() PCl3(g) ��H=-306 kJ?mol-1��
PCl3(g) ��H=-306 kJ?mol-1��
��2����H=������������-��Ӧ������������ӦPCl3(g)+Cl2(g)![]() PCl5(g) ��H=-93 kJ?mol-1���ʷ�ӦPCl5(g)
PCl5(g) ��H=-93 kJ?mol-1���ʷ�ӦPCl5(g)![]() PCl3(g)+Cl2(g) ��H=+93 kJ?mol-1��
PCl3(g)+Cl2(g) ��H=+93 kJ?mol-1��
�ֽ���=![]() ��100%=25%��
��100%=25%��
���ڷ�ӦPCl5![]() PCl3+Cl2�����ȷ�Ӧ�������¶ȣ�ƽ��������Ӧ�����ƶ����ֽ���������2���ڦ�1��
PCl3+Cl2�����ȷ�Ӧ�������¶ȣ�ƽ��������Ӧ�����ƶ����ֽ���������2���ڦ�1��
��3��3Cl2+2P![]() 2PCl3��PCl3+Cl2
2PCl3��PCl3+Cl2![]() PCl5������Ӧ��Ϊ���ȷ�Ӧ�������¶ȣ�������ƽ���������ƶ��������PCl3��ת���ʣ�ͬʱ��ֹPCl5�ֽ⡣
PCl5������Ӧ��Ϊ���ȷ�Ӧ�������¶ȣ�������ƽ���������ƶ��������PCl3��ת���ʣ�ͬʱ��ֹPCl5�ֽ⡣
��4�����ݸ�˹���ɣ�P��Cl2��������Ӧ��һ����Ӧ����PCl5�Ħ�HӦ������ȵġ�
��5��PCl5��H2O��Ӧ���ɵ��������H3PO4��HCl��HClO����PCl5![]() H3PO4��PԪ�ػ��ϼ�û�䣬��ClԪ�ػ��ϼ�Ҳ���䣬���÷�Ӧ���ɵ���������H3PO4��HCl�����Ը÷�Ӧ�Ļ�ѧ����ʽ��PCl5+4H2O
H3PO4��PԪ�ػ��ϼ�û�䣬��ClԪ�ػ��ϼ�Ҳ���䣬���÷�Ӧ���ɵ���������H3PO4��HCl�����Ը÷�Ӧ�Ļ�ѧ����ʽ��PCl5+4H2O![]() H3PO4+5HCl��
H3PO4+5HCl��

 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д� ����P(s)��Cl2(g)������Ӧ����PCl3(g)��PCl5(g)����Ӧ���̺�������ϵ��ͼ��ʾ(ͼ�еġ�H��ʾ����1mol���������)��
����P(s)��Cl2(g)������Ӧ����PCl3(g)��PCl5(g)����Ӧ���̺�������ϵ��ͼ��ʾ(ͼ�еġ�H��ʾ����1mol���������)��


