��Ŀ����
�������ӷ���ʽ��д��ȷ����( )
| A������ɫ�ĵ�����Һ��ͨ������SO2�����ɫ��Һ��I2+SO2+2H2O=2I-+SO32-+4H+ |
| B�������ʵ��������������������������Һ��ϣ�NH4++HSO3-+2OH-=SO32-+NH3��+2H2O |
C��̼����þ��Һ�м������ʯ��ˮ��Mg2++2HCO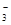 +Ca2++2OH-=CaCO3��+2H2O+MgCO3�� +Ca2++2OH-=CaCO3��+2H2O+MgCO3�� |
D��̼��������Һ�м����������������Һ��HCO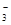 +Ba2++OH-=BaCO3��+H2O +Ba2++OH-=BaCO3��+H2O |
D
�ⵥ�ʾ��������ԣ��ܰ�SO2�����������ᣬ����ʽΪI2+SO2+2H2O=2I-+SO42-+4H+��B���������Ʋ��㣬������Ӧ���������ƺ�������泥�����ʽΪHSO3-+OH-=SO32- +2H2O��������þ��̼��þ���ܽ��С������C����������������þ������ʽΪMg2++HCO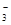 +Ca2++3OH-=CaCO3��+H2O+Mg(OH)2����������ȷ�Ĵ���D��
+Ca2++3OH-=CaCO3��+H2O+Mg(OH)2����������ȷ�Ĵ���D��
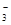 +Ca2++3OH-=CaCO3��+H2O+Mg(OH)2����������ȷ�Ĵ���D��
+Ca2++3OH-=CaCO3��+H2O+Mg(OH)2����������ȷ�Ĵ���D��
��ϰ��ϵ�д�
 Сѧ��10���ӿ������100��ϵ�д�
Сѧ��10���ӿ������100��ϵ�д�
�����Ŀ
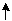
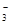 +OH
+OH ==CO
==CO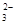 +H2O
+H2O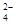 +Ba2+ ="=" BaSO4��
+Ba2+ ="=" BaSO4�� 2H++Cl��+ClO��
2H++Cl��+ClO��