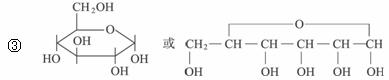
题目内容
“C1化学”是指以分子中只含一个碳原子的物质为原料进行物质合成的化学。“C1化学”对于缓解日益 严重的能源危机、合理利用煤和天然气等化石燃料、保护环境等都有非常重要的意义。合成气(CO+H2)是 “C1化学”中的常用原料。
(1)煤气化可生成合成气,该反应的化学方程式为____________________,用该方法生产合成气的一个典型缺陷是___________。
(2)将甲烷氧化可制合成气:CH4(g)+1/2O2(g) CO(g)+2H2(g) △H=-35.6 kJ/mol。该反应是_____(填“自发”或“非自发”)反应。
CO(g)+2H2(g) △H=-35.6 kJ/mol。该反应是_____(填“自发”或“非自发”)反应。
(3)通过乙醇制取合成气具有良好的应用前景。由乙醇制取合成气有如下两条路线:
a.水蒸气催化重整:CH3CH2OH(g)+H2O(g)→4H2(g)+2CO(g) △H=+255.58 kJ/mol
b.部分催化氧化:CH3CH2OH(g)+1/2O2(g)→ 3H2(g)+2CO(g) △H=+13.76 kJ/mol
下列说法错误的是____。
A.从原料消耗的角度来看,a路线制氢更有价值
B.从能量消耗的角度来看,b路线制氢更加有利
C.a路线制氢由于要消耗很多能量,所以在实际生产中意义不大
D.在以上两个反应中,原子利用率较高的是b反应
(4)工业用合成气制备二甲醚的生产流程如下:

催化反应室中(压力2.0~ 10.0 MPa,温度230~ 280℃)进行下列反应:
CO(g)+2H2(g) CH3OH(g) △H=-90.7 kJ/mol ①
CH3OH(g) △H=-90.7 kJ/mol ①
2CH3OH(g) CH3OCH3(g)+H2O(g) △H = -23.5 kJ/mol ②
CH3OCH3(g)+H2O(g) △H = -23.5 kJ/mol ②
CO(g) +H2O(g) CO2(g)+H2(g) △H=-41.2 kJ/mol ③
CO2(g)+H2(g) △H=-41.2 kJ/mol ③
①催化反应室中总反应3CO(g)+3H2(g) CH3OCH3(g)+CO2(g)的△H=_____。830℃ 时反应③的K=1.0,则在催化反应室中反应③的K_______(填“>”、“<”或“=”)1.0。
CH3OCH3(g)+CO2(g)的△H=_____。830℃ 时反应③的K=1.0,则在催化反应室中反应③的K_______(填“>”、“<”或“=”)1.0。
②上述流程中,可以循环使用的物质有____________。
CO(g)+2H2(g)
 CH3OH(g) △H=-90.7 kJ/mol ①
CH3OH(g) △H=-90.7 kJ/mol ①2CH3OH(g)
 CH3OCH3(g)+H2O(g) △H = -23.5 kJ/mol ②
CH3OCH3(g)+H2O(g) △H = -23.5 kJ/mol ②CO(g) +H2O(g)
 CO2(g)+H2(g) △H=-41.2 kJ/mol ③
CO2(g)+H2(g) △H=-41.2 kJ/mol ③ ①催化反应室中总反应3CO(g)+3H2(g)
 CH3OCH3(g)+CO2(g)的△H=_____。830℃ 时反应③的K=1.0,则在催化反应室中反应③的K_______(填“>”、“<”或“=”)1.0。
CH3OCH3(g)+CO2(g)的△H=_____。830℃ 时反应③的K=1.0,则在催化反应室中反应③的K_______(填“>”、“<”或“=”)1.0。 ②上述流程中,可以循环使用的物质有____________。
(1)C+H2O(g) CO+H2;能耗大
CO+H2;能耗大
(2)自发
(3)D
(4)①-246.1 kJ/mol;>;②CO、H2、甲醇和水
 CO+H2;能耗大
CO+H2;能耗大(2)自发
(3)D
(4)①-246.1 kJ/mol;>;②CO、H2、甲醇和水

练习册系列答案
 备战中考寒假系列答案
备战中考寒假系列答案
相关题目
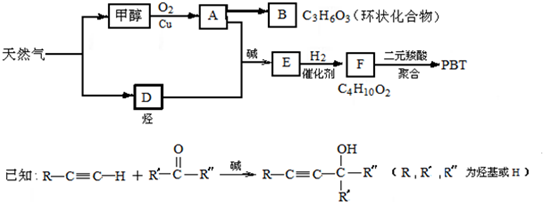
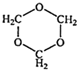

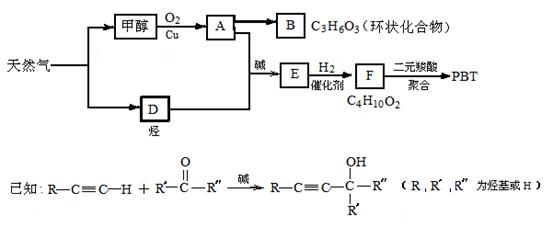
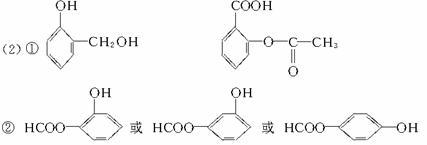 写出生成A的化学方程式:_________________________________。
写出生成A的化学方程式:_________________________________。