��Ŀ����
A��B��C��D���ֶ�����Ԫ�أ�A��B��Cͬ���ڣ�A��ԭ�Ӱ뾶��ͬ���������ģ�B��Dͬ���塣��֪DԪ�ص�һ�ֵ������ճ���������ˮ�����õ���������CԪ�صĵ��ʿ��Դ�A��B��Ԫ����ɵĻ������ˮ��Һ���û���BԪ�صĵ��ʡ�
��1��CԪ�������ڱ��е�λ�� ��
��2��AԪ����ˮ��Ӧ�����ӷ���ʽ�� ��
��
��3��д��CԪ�صĵ��ʴ�A��B��Ԫ����ɵĻ������ˮ��Һ���û���BԪ�صĵ��ʵĻ�ѧ����ʽ ��
��
��4��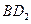 ��
��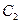 ������Ư���ԣ����ߵ�Ư��ԭ�� �������ͬ����ͬ����
������Ư���ԣ����ߵ�Ư��ԭ�� �������ͬ����ͬ����
��5�������Ǻϳɰ�����Ҫԭ�ϣ��ϳɰ���Ӧ���Ȼ�ѧ����ʽ���£�
3H2��N 2
2  2NH3��H=��92��4kJ��mol��1
2NH3��H=��92��4kJ��mol��1
�ٵ��ϳɰ���Ӧ�ﵽƽ��ı�ijһ������� ����
�ı�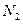 ��
�� ��
�� ����������Ӧ������ʱ��Ĺ�
����������Ӧ������ʱ��Ĺ�
ϵ����ͼ��ʾ��ͼ��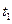 ʱ����ƽ���ƶ�����������
ʱ����ƽ���ƶ�����������
�� �����б�ʾƽ�������� �ĺ�����
�����
�ߵ�һ��ʱ���� ��

���¶�ΪT��ʱ����2a mol ��a mol
��a mol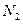 ����0��5 L �ܱ������У���ַ�Ӧ����
����0��5 L �ܱ������У���ַ�Ӧ����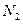 ��ת����Ϊ50������÷�Ӧ��ƽ�ⳣ��Ϊ ��
��ת����Ϊ50������÷�Ӧ��ƽ�ⳣ��Ϊ ��
��1��CԪ�������ڱ��е�λ�� ��
��2��AԪ����ˮ��Ӧ�����ӷ���ʽ��
 ��
����3��д��CԪ�صĵ��ʴ�A��B��Ԫ����ɵĻ������ˮ��Һ���û���BԪ�صĵ��ʵĻ�ѧ����ʽ
 ��
����4��
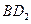 ��
��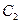 ������Ư���ԣ����ߵ�Ư��ԭ�� �������ͬ����ͬ����
������Ư���ԣ����ߵ�Ư��ԭ�� �������ͬ����ͬ������5�������Ǻϳɰ�����Ҫԭ�ϣ��ϳɰ���Ӧ���Ȼ�ѧ����ʽ���£�
3H2��N
 2
2  2NH3��H=��92��4kJ��mol��1
2NH3��H=��92��4kJ��mol��1�ٵ��ϳɰ���Ӧ�ﵽƽ��ı�ijһ������� ����
�ı�
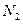 ��
�� ��
�� ����������Ӧ������ʱ��Ĺ�
����������Ӧ������ʱ��Ĺ�ϵ����ͼ��ʾ��ͼ��
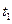 ʱ����ƽ���ƶ�����������
ʱ����ƽ���ƶ������������� �����б�ʾƽ��������
 �����
�ĺ������ߵ�һ��ʱ���� ��

���¶�ΪT��ʱ����2a mol
 ��a mol
��a mol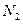 ����0��5 L �ܱ������У���ַ�Ӧ����
����0��5 L �ܱ������У���ַ�Ӧ����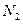 ��ת����Ϊ50������÷�Ӧ��ƽ�ⳣ��Ϊ ��
��ת����Ϊ50������÷�Ӧ��ƽ�ⳣ��Ϊ ����1���������ڣ���VIIA�� ��2�� 2Na+2H2O=2Na++2OH-+H2��
��3�� Cl2+Na2S=2NaCl+S�� ��4����ͬ ��5����ѹ t2-t3 4/a2
��3�� Cl2+Na2S=2NaCl+S�� ��4����ͬ ��5����ѹ t2-t3 4/a2
��

��ϰ��ϵ�д�
 �����ҵ���������ͯ������ϵ�д�
�����ҵ���������ͯ������ϵ�д�
�����Ŀ
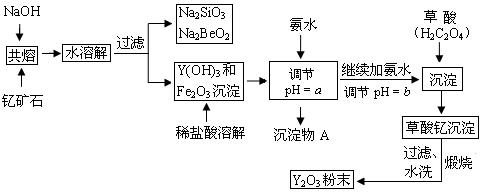
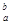 X
X
 �� ���ڹ�ҵ�Ϸ�Ӧ������___________�����豸���н��еģ���ʵ������________����ҩƷ��������D�Ĵ��ڣ���D��β��ֱ���ŷŵ���������ɻ�����Ⱦ�ĺ������������ ����
�� ���ڹ�ҵ�Ϸ�Ӧ������___________�����豸���н��еģ���ʵ������________����ҩƷ��������D�Ĵ��ڣ���D��β��ֱ���ŷŵ���������ɻ�����Ⱦ�ĺ������������ ����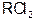 ����������Һʱ��Ϊ�˵õ�������Һ����ֹ���ʣ�������Һ�м�������R�ĵ��ʺ����ᡣ��֪
����������Һʱ��Ϊ�˵õ�������Һ����ֹ���ʣ�������Һ�м�������R�ĵ��ʺ����ᡣ��֪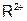 ��5�����Ӳ㣬���������2�����ӣ��������ƶ���ȷ����
��5�����Ӳ㣬���������2�����ӣ��������ƶ���ȷ����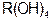 ��ǿ��
��ǿ��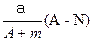 mol
mol  mol
mol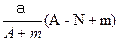 mol
mol mol
mol