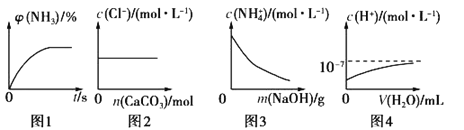
ЬтФПФкШн
ЁОЬтФПЁПШчЭМЫљЪОЃЌaжаЗХжУИНзХбѕЛЏЭЗлФЉЕФЪЏУоШоЃЌЯђaжаГжајЭЈШыЦјЬЌЮяжЪXЃЌПЩвдЙлВьЕНЪЏУоШоЩЯКкЩЋЗлФЉБфГЩКьЩЋЙЬЬЌЮяжЪЃЌЭЌЪБcДІЕФUаЮЙмжагаЮоЩЋвКЬхЩњГЩ(МйЩшXЦјЬхШЋВПЗДгІЃЌИїДІЗДгІОљЭъШЋ)ЁЃ

(1)XЪЧ_______
AЃЎH2 BЃЎCO CЃЎH2КЭCO DЃЎCH3CH2OHеєЦј
(2)аДГі a ДІЗДгІЕФЛЏбЇЗНГЬЪН:_______ЁЃ
(3)cДІвКЬхжївЊГЩЗжЕФНсЙЙМђЪНЮЊ______ЃЛ МьбщИУЮяжЪЕФВйзїВНжшЪЧ_______ЃЌЦфЯжЯѓЮЊ______ЁЃ
ЁОД№АИЁПD CH3CH2OH+CuO![]() CH3CHOЃЋCuЃЋH2O CH3 CHO ШЁЩйСПcДІЕФвКЬхгкЪдЙмжаЃЌМгШыаТжЦЧтбѕЛЏЭаќзЧвКЃЌМгШШ газЉКьЩЋГСЕэЩњГЩ
CH3CHOЃЋCuЃЋH2O CH3 CHO ШЁЩйСПcДІЕФвКЬхгкЪдЙмжаЃЌМгШыаТжЦЧтбѕЛЏЭаќзЧвКЃЌМгШШ газЉКьЩЋГСЕэЩњГЩ
ЁОНтЮіЁП
H2ЁЂCOКЭCH3CH2OHеєЦјЖМПЩвдЪЙaжаЕФКкЩЋЗлФЉ(бѕЛЏЭ)БфГЩКьЩЋЮяжЪ(Э)ЃЌЕЋH2КЭCOБЛбѕЛЏКѓЕФВњЮяЗжБ№ЪЧH2OКЭCO2ЃЌЫќУЧдкЭЈЙ§МюЪЏЛвКѓОљБЛЮќЪеЃЌcДІЕФUаЮЙмжаВЛЛсгаЮоЩЋвКЬхЩњГЩЃЌвђДЫЃЌXжЛФмЪЧCH3CH2OHеєЦјЃЌaДІЗЂЩњЕФЗДгІЮЊCH3CH2OHЃЋCuO![]() CH3CHOЃЋCuЃЋH2OЃЌbДІМюЪЏЛвЮќЪеЫЎЗжЃЌc ДІЕФ U аЮЙмжагаЮоЩЋвКЬхЮЊCH3CHOЃЌОнДЫЗжЮіПЩЕУНсТлЁЃ
CH3CHOЃЋCuЃЋH2OЃЌbДІМюЪЏЛвЮќЪеЫЎЗжЃЌc ДІЕФ U аЮЙмжагаЮоЩЋвКЬхЮЊCH3CHOЃЌОнДЫЗжЮіПЩЕУНсТлЁЃ
(1)ИљОнЗжЮіЃЌH2ЁЂCOКЭCH3CH2OHеєЦјЖМПЩвдЪЙaжаЕФКкЩЋЗлФЉ(бѕЛЏЭ)БфГЩКьЩЋЮяжЪ(Э)ЃЌЩњГЩЮяШєЮЊH2OКЭCO2ЛсБЛМюЪЏЛвЮќЪеЃЌUаЭЙмжаЮоЩЋвКЬхВЛЪЧЫЎЃЌЫљвдXПЩФмЪЧDЃЛ
(2)ИљОнЗжЮіЃЌaДІЗДгІЕФЛЏбЇЗНГЬЪНЃКCH3CH2OHЃЋCuO![]() CH3CHOЃЋCuЃЋH2OЃЛ
CH3CHOЃЋCuЃЋH2OЃЛ
(3)ИљОнЗжЮіЃЌcДІвКЬхжївЊГЩЗжЕФНсЙЙМђЪНЮЊCH3CHOЃЌМьбщИУЮяжЪЕФВйзїВНжшЪЧШЁЩйСПcДІЕФвКЬхгкЪдЙмжаЃЌМгШыаТжЦЧтбѕЛЏЭаќзЧвКЃЌМгШШЃЛЗЂЩњЕФЗДгІЛЏбЇЗНГЬЪНЮЊCH3CHOЃЋ2Cu(OH)2![]() CH3COOHЃЋCu2OЁ§ЃЋ2H2OЃЌЦфЯжЯѓЮЊгазЉКьЩЋГСЕэЩњГЩЁЃ
CH3COOHЃЋCu2OЁ§ЃЋ2H2OЃЌЦфЯжЯѓЮЊгазЉКьЩЋГСЕэЩњГЩЁЃ