��Ŀ����
��10�֣���ij2 L���ܱ������м���0.1 mol�ⵥ�ʺ�������(W)����943 Kʱ��������ӦW(s)��I2(g)  WI2(g)����I2��ת����Ϊ20%ʱ���ﻯѧƽ��״̬��
WI2(g)����I2��ת����Ϊ20%ʱ���ﻯѧƽ��״̬��
��1����Ӧ�ӿ�ʼ��ƽ��״̬��������ʱ20 s����ƽ����Ӧ����v(WI2)Ϊ________��
��2��943 Kʱ���÷�Ӧ��ƽ�ⳣ��K��________��
��3������ʼʱ������ĵ�Ϊ0.2 mol(������������)��ƽ��ʱ����������������
V(I2)��V(WI2)��________��
��4��������(1)ƽ��״̬�£�ͬʱ����I2(g)��WI2(g)��0.02 mol(������������)����ѧƽ����________(�����Ӧ�����淴Ӧ��)�����ƶ����жϵ�������______________________��
���𰸡�
(1)5��10��4mol��L��1��s��1 (2)0.25��(3)4��1��(4)�淴Ӧ��Qc��0.4>K��0.25
��������

��ϰ��ϵ�д�
 Ӧ�����������Ĵ���ѧ������ϵ�д�
Ӧ�����������Ĵ���ѧ������ϵ�д�
�����Ŀ
 WI2(g)����I2��ת����Ϊ20%ʱ���ﻯѧƽ��״̬��
WI2(g)����I2��ת����Ϊ20%ʱ���ﻯѧƽ��״̬��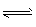 WI2(g)����I2��ת����Ϊ20%ʱ���ﻯѧƽ��״̬��
WI2(g)����I2��ת����Ϊ20%ʱ���ﻯѧƽ��״̬��