��Ŀ����
����Ŀ�������й���Һ������Ũ�ȵĹ�ϵʽ�У���ȷ����
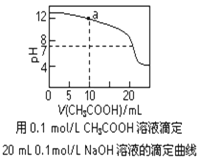
A. pH��ͬ�Ģ�CH3COONa����NaHCO3����Na2CO3������Һ�е�c(Na��)���ۣ��ڣ���
B. 0.1mol��L��1ij��Ԫ����ǿ����NaHA��Һ�У�c(Na+)=2c(A2-)��c(HA-)��c(H2A)
C. ͼ��pH��7ʱ��c(Na��)��c(CH3COO��) ��c(OH��)��c(H��)
D. ͼ��a����Һ�и�����Ũ�ȵĹ�ϵ�ǣ�c(OH��)��c(H��)��c(CH3COO��)��2c(CH3COOH)
���𰸡�D
��������
A��CH3COONa��NaHCO3��Na2CO3ˮ��̶������������Ե�������Һ��pH��ͬʱ�������ʵ���Ũ���ɴ�С��˳���Ǣۣ��ڣ��٣���A����
B�����������غ㣬0.1mol��L��1ij��Ԫ����ǿ����NaHA��Һ�У�c(Na+)=c(A2-)��c(HA-)��c(H2A)����B����
C�����ݵ���أ�ͼ��pH��7ʱ��c(Na��)=c(CH3COO��)��c(OH��)��c(H��)����C����
D�� ͼ��a����Һ�к��е�Ũ�ȵĴ����ƺ��������ƣ����������غ㣺c(Na��)= 2c(CH3COO��)��2c(CH3COOH)�����ݵ���غ�c(Na��)+ c(H��)=c(CH3COO��) +c(OH��)��������ʽ�ɵã�c(OH��)��c(H��)��c(CH3COO��)��2c(CH3COOH)����D��ȷ��
��ѡD��

 ��ʦָ����ĩ��̾�ϵ�д�
��ʦָ����ĩ��̾�ϵ�д�