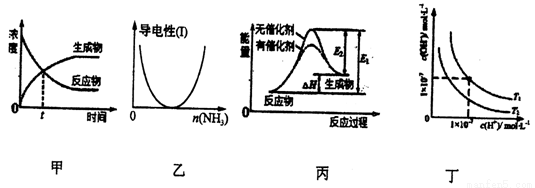
��Ŀ����
����ָ����Ӧ�����ӷ���ʽ��ȷ����
A. �����м���Na2CO3��Һ�У�CO32-+C6H5OH��C6H5O-+HCO3-
B. 0.3molFeI2��0.4molCl2����Һ�з�Ӧ��6Fe2++10I-+8Cl2=6Fe3++16Cl-+5I2
C. NaHSO4��Һ��Ba(OH)2��Һ��Ӧ����Һ�����ԣ�H++SO42-+Ba2++OH-=BaSO4��+H2O
D. AgCl��������������ˮ��Ag++2NH3��H2O=[Ag(NH3)2]++2H2O
��ϰ��ϵ�д�
�����Ŀ



 ��BrCH2COOC2H5Ϊԭ���Ʊ�
��BrCH2COOC2H5Ϊԭ���Ʊ� �ĺϳ�·������ͼ �����Լ�����ѡ�����ϳ�·������ͼʾ�����£�
�ĺϳ�·������ͼ �����Լ�����ѡ�����ϳ�·������ͼʾ�����£�  CH3COOH
CH3COOH CH3COOCH2CH3
CH3COOCH2CH3