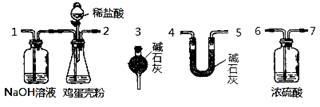
��Ŀ����
��12�֣��������к��д����ơ�����þ�ͼص�Ԫ�ء�ʵ���ҿ���ͼʾ���̲ⶨ�������иơ�þԪ�غ��������ݵķ�Ӧ���Ա�ʾΪ��Ca2++Y2-=CaY��Mg2++Y2-=MgY���ش��������⡣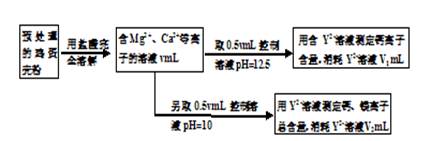
��1���ⶨCa2+��Mg2+�ܺ���ʱ��������ҺpH=10����pH���ⶨ����� ���ƫ����ƫС������Ӱ�족����
��2���ⶨCa2+�����Ĺؼ��ǿ�����Һ��pH��ʹ��Һ��Mg2+�γɳ�������Ҫʹ��Һ��c(Mg2+)������1.2��10-7mol/L������ҺpHӦ��С�� (��֪��Ksp[Mg(OH)2]=1.2��10��11������ʵ����������½���)��
��3����������Ƿ�����Ϊmg����Һ��Y2+Ũ��Ϊc mol/L��������Ʒ��þԪ������������ ��
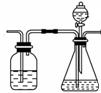
��4����ͬѧ����ɲ�������װ�á�ͨ���ⶨ�����������ᷴӦ�����Ķ�����̼�������ܲⶨ�������еĸƵ��ܺ�����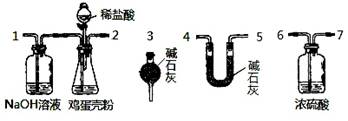
�ٲ��ø÷�������ʵ�飬��װ�ô����ҵ�����˳�����Σ�
1��2��__ ________________�����װ�ö�Ӧ�ӿڵ�������ţ���
�����ų�ʵ�������Ͳ�����Ӱ�����أ���ʵ�鷽����õĽ���Ƿ�ȷ��
��________���ȷ��������һ��ȷ�������жϡ�����
ԭ����___________________ __
��
��12�֣���1��ƫС��2�֣� ��2��12 ��2�֣� ��3�� ��3�֣�
��3�֣�
��4����6��7��4��5��3��6��7��5��4��3��2�֣��ڲ�һ��ȷ��
��1�֣���������и�Ԫ��ȫ����̼�����ʽ���ڣ��Ҳ��ٺ����������ᷴӦ��������������������ʣ���ⶨ���ȷ������ȷ��
�����������⣬ֻҪ������������֣���2�֣�
����

 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�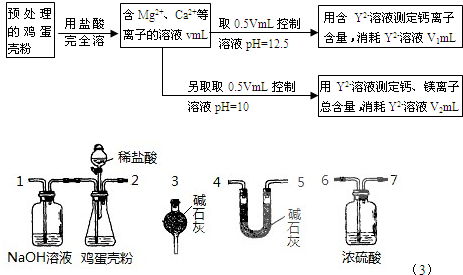

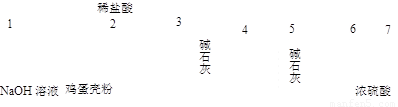



 ��________���ȷ��������һ��ȷ�������жϡ�����
��________���ȷ��������һ��ȷ�������жϡ����� ��
��