��Ŀ����
һ���¶ȷ�Χ�����Ȼ����۽��س�ʯ����Ҫ�ɷ�ΪKAlSi3O8�����Ƶ��Ȼ��أ���Ҫ��Ӧ�ǣ�
NaCl��l��+KAlSi3O8��s��?KCl��l��+NaAlSi3O8��s����
��1��������Ӧ�漰�ĵ�������Ԫ���У����Ӱ뾶��С���� ����Ԫ�ط��ű�ʾ����Clԭ����Siԭ�ӿ����γɵ���˷��ӣ��仯ѧ�����ͼ��Ƕ���ȣ��������Ϊ ���ӣ�����ԡ��Ǽ��ԡ�����
��2����ʾԭ�ӻ����ӽṹ�Ļ�ѧ�����У�ԭ�ӽṹʾ��ͼ����������Ų�ʽ�������ʾʽ������ѡ�����꾡������������˶�״̬�ķ�ʽ������ʾ�����Ӻ�����ӵ��˶�״̬�� ��
��3������Ԫ�ص�����������Ӧˮ�����У�������ǿ��������ǿ�Ӧ����ԭ�����ڣ� ���õ��뷽��ʽ��ʾ����
��4��Al��Cl�����γɷ��Ӿ���AlCl3������AlCl3��Һ��˵��������� ��
A��ˮ��Һ������Ũ�ȴ�С��c��Cl-����c��Al3+����c��H+����c��OH-��
B�������¶ȣ�AlCl3ˮ��ҺpH��С��c��Al3+����С
C��c��Al3+��+c��H+��=c��Cl-��+c��OH-��
D��c��Cl-��=3c��Al3+��+3c��AlO2-��
��5���������ڱȽ�Na��Al���������ǿ������ʵ�� ��
A������������Ӧˮ����ļ��� B��Na��AlCl3��Һ��Ӧ
C��������H2O��Ӧ�����׳̶� D���Ƚ�ͬŨ��NaCl��AlCl3��pHֵ
��6��Na��O2��Ӧ�γ�Na2O��Na2O2�Ļ����������ӵĸ�����Ϊ ���õ���ʽ��ʾNa2O2���γɹ��̣� ��
NaCl��l��+KAlSi3O8��s��?KCl��l��+NaAlSi3O8��s����
��1��������Ӧ�漰�ĵ�������Ԫ���У����Ӱ뾶��С����
��2����ʾԭ�ӻ����ӽṹ�Ļ�ѧ�����У�ԭ�ӽṹʾ��ͼ����������Ų�ʽ�������ʾʽ������ѡ�����꾡������������˶�״̬�ķ�ʽ������ʾ�����Ӻ�����ӵ��˶�״̬��
��3������Ԫ�ص�����������Ӧˮ�����У�������ǿ��������ǿ�Ӧ����ԭ�����ڣ�
��4��Al��Cl�����γɷ��Ӿ���AlCl3������AlCl3��Һ��˵���������
A��ˮ��Һ������Ũ�ȴ�С��c��Cl-����c��Al3+����c��H+����c��OH-��
B�������¶ȣ�AlCl3ˮ��ҺpH��С��c��Al3+����С
C��c��Al3+��+c��H+��=c��Cl-��+c��OH-��
D��c��Cl-��=3c��Al3+��+3c��AlO2-��
��5���������ڱȽ�Na��Al���������ǿ������ʵ��
A������������Ӧˮ����ļ��� B��Na��AlCl3��Һ��Ӧ
C��������H2O��Ӧ�����׳̶� D���Ƚ�ͬŨ��NaCl��AlCl3��pHֵ
��6��Na��O2��Ӧ�γ�Na2O��Na2O2�Ļ����������ӵĸ�����Ϊ
��������1���漰������Ԫ����Na��Al��Si��Cl�����Ӱ뾶��С��ΪAl�����ӽṹ�Գ�ʱΪ�Ǽ��Է��ӣ�
��2����ʾ�����Ӻ�����ӵ��˶�״̬��Ϊ�����ʾʽ��
��3������ǿ��������ǿ�ӦΪ����������Ϊ�����������
��4��AlCl3Ϊǿ�������Σ�ˮ������ԣ���ϵ���غ㡢�����غ�����⣻
��5���ȽϽ�����ǿ�����ɸ��ݶ�Ӧ������������ˮ����ļ��ԡ��������ᡢˮ��Ӧ�ľ��ҳ̶ȵȽǶȽ����жϣ�
��6��Na2O2�е�������ΪO22-���Դ��ж�Na2O��Na2O2�Ļ����������ӵĸ����ȣ�
��2����ʾ�����Ӻ�����ӵ��˶�״̬��Ϊ�����ʾʽ��
��3������ǿ��������ǿ�ӦΪ����������Ϊ�����������
��4��AlCl3Ϊǿ�������Σ�ˮ������ԣ���ϵ���غ㡢�����غ�����⣻
��5���ȽϽ�����ǿ�����ɸ��ݶ�Ӧ������������ˮ����ļ��ԡ��������ᡢˮ��Ӧ�ľ��ҳ̶ȵȽǶȽ����жϣ�
��6��Na2O2�е�������ΪO22-���Դ��ж�Na2O��Na2O2�Ļ����������ӵĸ����ȣ�
����⣺��1���漰������Ԫ����Na��Al��Si��Cl�����Ӱ뾶��С��ΪAl��Clԭ����Siԭ�ӿ����γɵ���˷��ӣ��仯ѧ�����ͼ��Ƕ���ȣ�����Ϊ��������ṹ��Ϊ�Ǽ��Է��ӣ��ʴ�Ϊ��Al���Ǽ��ԣ�
��2��������ԭ�Ӻ�����10�����ӣ������ʾʽΪ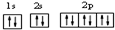 ���ʴ�Ϊ��
���ʴ�Ϊ�� ��
��
��3������ǿ��������ǿ�ӦΪ����������Ϊ������������ɷ�����ʽ����ͼ�ʽ���룬ΪH++AlO2-+H2O?Al��OH��3?Al3++3OH-��
�ʴ�Ϊ��H++AlO2-+H2O?Al��OH��3?Al3++3OH-��
��4��A��AlCl3Ϊǿ�������Σ�ˮ������ԣ���ˮ��Һ������Ũ�ȴ�СΪ��c��Cl-����c��Al3+����c��H+����c��OH-������A��ȷ��
B�������¶ȣ��ٽ�AlCl3��ˮ�⣬����ҺpH��С��c��Al3+����С����B��ȷ��
C���ɵ���غ��֪3c��Al3+��+c��H+��=c��Cl-��+c��OH-������C����
D���������غ��֪ӦΪc��Cl-��=3c��Al3+��+3c��Al��OH��3������D����
�ʴ�Ϊ��CD��
��5���ȽϽ�����ǿ�����ɸ��ݶ�Ӧ������������ˮ����ļ��ԡ��������ᡢˮ��Ӧ�ľ��ҳ̶ȵȽǶȣ��Ʋ��ܴ���Һ���û��������������ڱȽϽ�����ǿ����
�ʴ�Ϊ��B��
��6��Na2O2�е�������ΪO22-�����ж�Na2O��Na2O2�Ļ����������ӵĸ�����Ϊ1��2���õ���ʽ��ʾNa2O2���γɹ���Ϊ ��
��
�ʴ�Ϊ��1��2�� ��
��
��2��������ԭ�Ӻ�����10�����ӣ������ʾʽΪ
 ���ʴ�Ϊ��
���ʴ�Ϊ�� ��
����3������ǿ��������ǿ�ӦΪ����������Ϊ������������ɷ�����ʽ����ͼ�ʽ���룬ΪH++AlO2-+H2O?Al��OH��3?Al3++3OH-��
�ʴ�Ϊ��H++AlO2-+H2O?Al��OH��3?Al3++3OH-��
��4��A��AlCl3Ϊǿ�������Σ�ˮ������ԣ���ˮ��Һ������Ũ�ȴ�СΪ��c��Cl-����c��Al3+����c��H+����c��OH-������A��ȷ��
B�������¶ȣ��ٽ�AlCl3��ˮ�⣬����ҺpH��С��c��Al3+����С����B��ȷ��
C���ɵ���غ��֪3c��Al3+��+c��H+��=c��Cl-��+c��OH-������C����
D���������غ��֪ӦΪc��Cl-��=3c��Al3+��+3c��Al��OH��3������D����
�ʴ�Ϊ��CD��
��5���ȽϽ�����ǿ�����ɸ��ݶ�Ӧ������������ˮ����ļ��ԡ��������ᡢˮ��Ӧ�ľ��ҳ̶ȵȽǶȣ��Ʋ��ܴ���Һ���û��������������ڱȽϽ�����ǿ����
�ʴ�Ϊ��B��
��6��Na2O2�е�������ΪO22-�����ж�Na2O��Na2O2�Ļ����������ӵĸ�����Ϊ1��2���õ���ʽ��ʾNa2O2���γɹ���Ϊ
 ��
���ʴ�Ϊ��1��2��
 ��
�����������⿼���Ϊ�ۺϣ��漰Ԫ�������ɡ�����Ũ�ȴ�С�Ƚϣ���ˮˮ���Ӧ�õ�֪ʶ��Ϊ��Ƶ���㣬��Ŀ���������߿����Ѷ����У�ע����ػ���֪ʶ�Ļ��ۣ�

��ϰ��ϵ�д�
 ��������һ���þ�ϵ�д�
��������һ���þ�ϵ�д� Сѧ��10����Ӧ����ϵ�д�
Сѧ��10����Ӧ����ϵ�д�
�����Ŀ
 ˮ������Ϊһ����Ҫ�ľ�ϸ����ԭ�ϣ���ũҩ��ҽҩ���л��ϳ����й㷺��;�� �����ط��Ʊ�ˮ���£��ɷ�Ϊ�����Σ���һ��Ϊ�����Ȼ��Σ��ڶ���Ϊ����ˮ��Σ��ܷ�Ӧ����ʽΪ����NH2��2CO+NaClO+2NaOH��H2N-NH2?H2O+NaCl+Na2CO3��
ˮ������Ϊһ����Ҫ�ľ�ϸ����ԭ�ϣ���ũҩ��ҽҩ���л��ϳ����й㷺��;�� �����ط��Ʊ�ˮ���£��ɷ�Ϊ�����Σ���һ��Ϊ�����Ȼ��Σ��ڶ���Ϊ����ˮ��Σ��ܷ�Ӧ����ʽΪ����NH2��2CO+NaClO+2NaOH��H2N-NH2?H2O+NaCl+Na2CO3��
