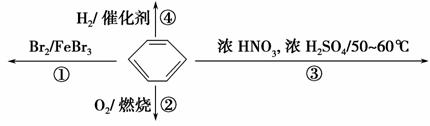
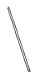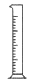��Ŀ����
��NA��ʾ�����ӵ���������ֵ������˵����ȷ����
A���ڱ�״���£�0.5molNO��0.5molO2��Ϻ����������Ϊ0.75 NA
B�����³�ѹ�£�14g��C2H4��C4H8��������к��е�ԭ����Ϊ3NA
C��0.1molAlCl3��ȫת��Ϊ�����������壬����0.l NA����������
D��1.0L 1.0 mol��L-1 Na2SiO3ˮ��Һ�к��е���ԭ����Ϊ3NA
B

����ʵ����������ﵽԤ��Ŀ�ĵ��� ( )
| ʵ��Ŀ�� | ���� | |
| A | ���Ƚ�ˮ���Ҵ����ǻ���Ļ����� | �ý����Ʒֱ���ˮ���Ҵ���Ӧ |
| B | ��֤��CH2=CHCHO�к���̼̼˫�� | �μ�����KMnO4��Һ�����Ϻ�ɫ�Ƿ���ȥ |
| C | ��ȷ���ס�����Ԫ�طǽ����Ե�ǿ�� | �ⶨͬ��ͬŨ�ȵ�Na3PO4��Na3AsO4ˮ��Һ��pH |
| D | ����ȥ���л��еı��� | ����Һ�м���NaOH��Һ����ַ�Ӧ��Һ |
ijʵ��С������0.10mol/LNaOH��Һ�������й�����ʵ�飬�ش��������⡣
��1����ʵ���д�ԼҪʹ��475mLNaOH��Һ��������Ҫ����NaOH���� g��
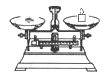
��2������ͼ��ѡ�����NaOH��������Ҫ�������ǣ�����ĸ�� ��
| ���� | ������ƽ �������룩 | С�ձ� | ����ǯ | ������ | ҩ�� | ��Ͳ |
| ���� | | | | | | |
| ��� | a | b | c | d | e | f |
��3�����������ʹ������ҺŨ��ƫ�͵��ǣ�����ţ�
�ٳ�������ʱ�����̸ߣ����̵� �����õ���������
����Һת�Ƶ�����ƿ��δ����ϴ�Ӳ��� ��ת����Һǰ����ƿ������������ˮ
�ݶ���ʱ����������ƿ�Ŀ̶���
�����ձ����ܽ�NaOH��������������Һע������ƿ��
�߶��ݺ�ҡ�ȣ�����Һ�潵�ͣ��ֲ�������ˮ�����´ﵽ�̶�ʵ����������
������Ͳ��ȡһ����Ũ��������ϡ���ᣬת�Ƶ��ձ���������ˮϴ����Ͳ
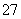 �ķ�ˮ���Խϴ�ij������ˮ�к�5.0��10��3 mol��L��1��Cr2O
�ķ�ˮ���Խϴ�ij������ˮ�к�5.0��10��3 mol��L��1��Cr2O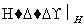 Cr3����Fe3��
Cr3����Fe3��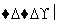 Cr(OH3)��Fe(OH)3
Cr(OH3)��Fe(OH)3 ��2.0��10��13 mol��L��1���������Cr3����Ũ��Ϊ________��
��2.0��10��13 mol��L��1���������Cr3����Ũ��Ϊ________��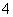 ��
��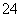 ��
��