��Ŀ����
ζ��(��Ҫ�ɷֵĻ�ѧʽΪC5H8NO4 Na��H2O)������������ʳ�Σ�ijѧ����ⶨijƷ��ζ���Ĵ��ȣ���������ʵ�飺
�ٳ�ȡijƷ�ƵĴ�װζ����Ʒ1.000 g�������ձ��У���������ˮ�ܽ⣬�ָ������£�
�����ձ��У������Թ�������ϡ�����ữ��A��Һ����ַ�Ӧ��ʹ������ȫ��
�۹��ˣ�������ˮ����ϴ�ӳ�����Σ�
�ܽ�������ɡ����������ù�������Ϊ0.368 g��
��������ʵ��������ش��������⣺
��1�������A��Һ��________����������A��Һ�Ƿ�����ķ�����_______________________��
��2������ϴ���DZز����ٵ�һ������������ϴ�ӣ���õĽ����_____________��
��3����ɳ���Ӧ���õķ�����________(����ĸ)��
A�����º�� B�����º�� C������̫����ɹ��
��4����Ʒ��ζ���Ĵ���(��ζ����C5H8NO4 Na��H2O����������)Ϊ__________��
��5��д��ζ����ʳ���ж����е�Ԫ�ص����ӽṹʾ��ͼ��___________��
 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д��±���ʾW��X��Y��ZΪ������Ԫ�أ�������Ԫ�ص�ԭ������������֮��Ϊ22������˵����ȷ���� �� ��
| X | Y |
|
W |
|
| Z |
A��X��Y��Z����Ԫ����ͼ��⻯��ķе���������
B����X��Y��������Ԫ���γɵĻ�������ֻ�й��ۼ�
C������WY2��W3X4��WZ4�����۵�ߡ�Ӳ�ȴ������
D��X�������ӱ�Y�����������Ӱ뾶��
������������һ���������ܹ��棬��������Ӧ�Լ���ᷢ����ѧ�仯�����������ӷ���ʽ��ȷ���ǣ� ��
ѡ�� | ������ | �����Լ� | �����Լ�������Ӧ�� |
A | Fe2����NO | ϡ���� | 3Fe 2����NO |
B | Fe3����I����ClO�� | ����������Һ | Fe3����3OH��= Fe(OH)3�� |
C | Ba2����HCO | ����������Һ | H |
D | Al3����Cl����NO | ��������������Һ | Al3����3OH��= Al(OH)3�� |



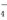 =X2����Z2��H2O(δ��ƽ)��
=X2����Z2��H2O(δ��ƽ)�� ���ӷ���ʽ
���ӷ���ʽ ��K��
��K�� CO
CO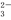 ��H2O
��H2O
