��Ŀ����
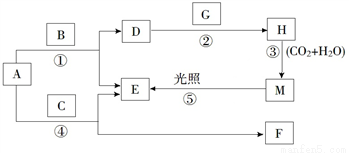
����A��B��C��D��E��F��G��H��M�������ʣ�����AΪ����ɫ��ĩ��GΪ����ɫ���嵥�ʣ�EΪ��ɫ���嵥�ʣ�F��ˮ��Һ��ʯ��ˮ��Ͽɵ�D����Һ������֮����ת����ϵ��ͼ��ʾ�����������ص������������ȥ��
��ش��������⣺
��1��д��G��H�Ļ�ѧʽ��G__________��H__________��
F��������_________________��A����Ҫ��;��________________��
��2��д����Ӧ�٢ڵ����ӷ���ʽ��
��____________________��
��____________________��
��3����֪CN��(�������))��Cl��������֮���������(CN)2��������Cl2������������֮�����ҳ��³�ѹ�������塣��д��MnO2��HCN��Һ���ȷ�Ӧ�Ļ�ѧ����ʽ��_______________��
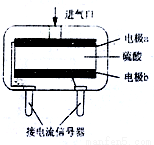
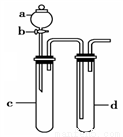
��4��������ͼװ�ý���ʵ�飬֤��������ǿ����KMnO4>Cl2>Br2��
��ѡ�Լ���KBr��Һ��KMnO4��Ũ���ᡢŨ���ᡣ
��֪��2KMnO4+16HCl(Ũ)=2KCl+ 2MnCl2+5Cl2��+8H2O
��ش��������⣺
��װ��a ��d��ʢ�ŵ��Լ��ֱ���__________��__________��
��ʵ���й۲쵽������Ϊ_____________________��
�۴�ʵ��װ�õIJ���֮����_____________________��
 ��ѧ��ʦ����ϵ�д�
��ѧ��ʦ����ϵ�д�

 ��
�� ˵������ȷ���ǣ�������
˵������ȷ���ǣ�������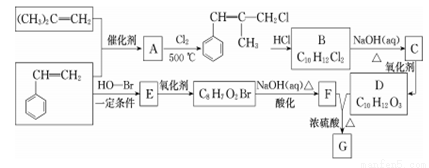
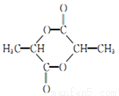 �ĺϳ�·�ߣ�_______��
�ĺϳ�·�ߣ�_______��
 2MgSO4(s)+N2(g) ��H =akJ/ mol����ƽ�ⳣ�����¶ȵĹ�ϵ��ͼ����ʾ��
2MgSO4(s)+N2(g) ��H =akJ/ mol����ƽ�ⳣ�����¶ȵĹ�ϵ��ͼ����ʾ��
 ��
�� ��
�� �����ʣ�ͨ�����¼���ʵ�鲽����Գ�ȥ��Щ���ʡ�
�����ʣ�ͨ�����¼���ʵ�鲽����Գ�ȥ��Щ���ʡ� �����Թ�����
�����Թ����� ��Һ��
��Һ�� �����Թ�����
�����Թ����� ��Һ��
��Һ�� �����Թ�����
�����Թ����� ��Һ��
��Һ�� ���ˣ�
���ˣ� ����ϡ�����������ݲ�������ȷ�IJ���˳���ǣ� ��
����ϡ�����������ݲ�������ȷ�IJ���˳���ǣ� ��