��Ŀ����
��֪�������ݣ�
ѧ����ʵ������ȡ������������Ҫ�������£�
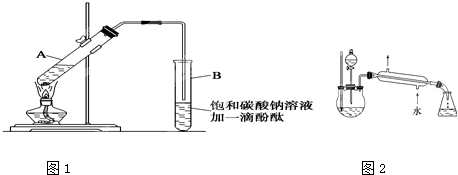
����30mL�Ĵ��Թ�A�а������2��3��2�ı�������Ũ���ᡢ�Ҵ�������Ļ����Һ��
�ڰ�ͼ1���Ӻ�װ�ã�װ�����������ã�����С����ȵؼ���װ�л����Һ�Ĵ��Թ�5-10min��
�۴��Թ�B�ռ���һ�����IJ����ֹͣ���ȣ���ȥ�Թ�B��������Ȼ���ô��ֲ㣻
�ܷ�������������㡢ϴ�ӡ����
�������ĿҪ��ش��������⣺
��1�����Ƹû����Һ����Ҫ��������Ϊ
��2������ʵ���б���̼������Һ�������ǣ�����ĸ��
A���к�������Ҵ�
B���к����Ტ���ղ����Ҵ�
C�����������ڱ���̼������Һ�е��ܽ�ȱ���ˮ�и�С�������ڷֲ�����
D�������������ɣ���������
��3�����������ҪС����ȼ��Ȳ���������Ҫ������
��4��ָ����������۲쵽������
A��P2O5B����ˮNa2SO4 C����ʯ�� D��NaOH����
��5��ij��ѧ����С���������ͼ2��ʾ����ȡ����������װ�ã�ͼ�е�����̨�����С�����װ������ȥ��������ͼװ����ȣ���װ�õ���Ҫ�ŵ���
| ���� | �۵㣨�棩 | �е㣨�棩 | �ܶȣ�g/cm3�� |
| �Ҵ� | -117.0 | 78.0 | 0.79 |
| ���� | 16.6 | 117.9 | 1.05 |
| �������� | -83.6 | 77.5 | 0.90 |
| Ũ���ᣨ98%�� | - | 338.0 | 1.84 |
����30mL�Ĵ��Թ�A�а������2��3��2�ı�������Ũ���ᡢ�Ҵ�������Ļ����Һ��
�ڰ�ͼ1���Ӻ�װ�ã�װ�����������ã�����С����ȵؼ���װ�л����Һ�Ĵ��Թ�5-10min��
�۴��Թ�B�ռ���һ�����IJ����ֹͣ���ȣ���ȥ�Թ�B��������Ȼ���ô��ֲ㣻
�ܷ�������������㡢ϴ�ӡ����

�������ĿҪ��ش��������⣺
��1�����Ƹû����Һ����Ҫ��������Ϊ
Ӧ�ȼ����Ҵ���Ȼ���ҡ���Թܱ���������Ũ���ᣬ�����������
Ӧ�ȼ����Ҵ���Ȼ���ҡ���Թܱ���������Ũ���ᣬ�����������
��д����ȡ���������Ļ�ѧ��Ӧ����ʽCH3COOH+CH3CH2OH
CH3COOCH2CH3+H2O
| Ũ���� |
| ���� |
CH3COOH+CH3CH2OH
CH3COOCH2CH3+H2O
��| Ũ���� |
| ���� |
��2������ʵ���б���̼������Һ�������ǣ�����ĸ��
BC
BC
��A���к�������Ҵ�
B���к����Ტ���ղ����Ҵ�
C�����������ڱ���̼������Һ�е��ܽ�ȱ���ˮ�и�С�������ڷֲ�����
D�������������ɣ���������
��3�����������ҪС����ȼ��Ȳ���������Ҫ������
���������Ҵ��Ļӷ������ٸ���Ӧ�ķ���
���������Ҵ��Ļӷ������ٸ���Ӧ�ķ���
����4��ָ����������۲쵽������
�Թ�B�е�Һ��ֳ��������㣬�ϲ���ɫ���²�Ϊ��ɫҺ�壬���²�Һ��ĺ�ɫ��dz
�Թ�B�е�Һ��ֳ��������㣬�ϲ���ɫ���²�Ϊ��ɫҺ�壬���²�Һ��ĺ�ɫ��dz
�����������������Ϊ�˸�������������ѡ�õĸ����Ϊ������ĸ��B
B
��A��P2O5B����ˮNa2SO4 C����ʯ�� D��NaOH����
��5��ij��ѧ����С���������ͼ2��ʾ����ȡ����������װ�ã�ͼ�е�����̨�����С�����װ������ȥ��������ͼװ����ȣ���װ�õ���Ҫ�ŵ���
���������¶ȼƣ����ڿ��Ʒ���װ���з�ӦҺ���¶ȣ����ٸ�����ķ������������˷�Һ©���������ڼ�ʱ���䷴Ӧ���Һ����������������IJ�����������������װ�ã��������ռ���������������
���������¶ȼƣ����ڿ��Ʒ���װ���з�ӦҺ���¶ȣ����ٸ�����ķ������������˷�Һ©���������ڼ�ʱ���䷴Ӧ���Һ����������������IJ�����������������װ�ã��������ռ���������������
����������1����Ũ��������ˮ���Ⱥͼ���ʱ�Թ���Һ�������������Թ��ݻ���1/3��������
��2������������ԣ��ܺͱ���̼������Һ��Ӧ���ѱ����գ����������ڱ���̼������Һ�е��ܽ�Ƚ�С��
��3���Ҵ�������ķе�ϵͣ��ӷ���
��4��������������Ժ��������������ڱ���̼���Ʒ�����
��5���Ӹ�װ���봫ͳ�Ʊ�װ�õ�ʹ�õ������IJ�ͬ���з�����
��2������������ԣ��ܺͱ���̼������Һ��Ӧ���ѱ����գ����������ڱ���̼������Һ�е��ܽ�Ƚ�С��
��3���Ҵ�������ķе�ϵͣ��ӷ���
��4��������������Ժ��������������ڱ���̼���Ʒ�����
��5���Ӹ�װ���봫ͳ�Ʊ�װ�õ�ʹ�õ������IJ�ͬ���з�����
����⣺��1���Ҵ��к�������ˮ��Ӧ��Ũ������뵽�Ҵ��У���ֹ�����ܽ�ʱ�ų��������ȵ���Һ��ɽ������⣬����ʱ���Թ���Һ�������������Թ��ݻ���1/3��������ҩƷ��˳��Ϊ��һ��30mL���Թ���ע��4mL�Ҵ����ٷֱ�����4mL���ᡢ1mLŨ���ᣨ�����Ũ����ļ���˳��ɻ��������ӱ����Թ�ʹ֮��Ͼ��ȣ�ҩƷ���������ܳ���10mL����������Ҵ������ķ�ӦΪ��CH3COOH+CH3CH2OH
CH3COOCH2CH3+H2O��
�ʴ�Ϊ��Ӧ�ȼ����Ҵ���Ȼ���ҡ���Թܱ���������Ũ���ᣬ����������CH3COOH+CH3CH2OH
CH3COOCH2CH3+H2O����
��2������������ԣ��ܺͱ���̼������Һ��Ӧ���ѱ����գ����������ڱ���̼������Һ�е��ܽ�Ƚ�С�����ڷ��룬
�ʴ�Ϊ��BC����
��3����Ӧ�����Ҵ�������ķе�ϵͣ����ô����ȣ�������Ӧ���������������ʧԭ�ϣ��¶ȹ������ܷ�����������Ӧ������Ϊ��ֹ�Ҵ�������ӷ������ԭ�ϵ���ʧ��ӦС����ȣ�
�ʴ�Ϊ�����������Ҵ��Ļӷ������ٸ���Ӧ�ķ�����
��4��̼����ˮ��ʼ��ԣ��������������ڱ���̼������Һ���ܶȱ�ˮС������ζ����ʱ�����̼���Ʒ�Ӧ��ʹ��Һ��ɫ��dz��������������������ˮ�����Ƴ�ȥ������ˮ����ˮ��������ˮ�γ������ƽᾧˮ�������ѡ��P2O5����ʯ�Һ�NaOH�ȹ����������Է��������������ԣ�P2O5��ˮ�����ᣩ�����������ˮ�⣬
�ʴ�Ϊ���Թ�B�е�Һ��ֳ��������㣬�ϲ���ɫ���²�Ϊ��ɫҺ�壬���²�Һ��ĺ�ɫ��dz��B����
��5���Ӹ�װ���봫ͳ�Ʊ�װ�õ�ʹ�õ������IJ�ͬ���з������������¶ȼƣ������ڿ��Ʒ���װ���з�ӦҺ���¶ȣ����ݷ�Ӧ�Ŀ������ص㣬���ӷ�Ӧ��������ƽ�����������ƶ���������������IJ���������������װ�ã��������ռ����
�ʴ�Ϊ�����������¶ȼƣ����ڿ��Ʒ���װ���з�ӦҺ���¶ȣ����ٸ�����ķ������������˷�Һ©���������ڼ�ʱ���䷴Ӧ���Һ����������������IJ�����������������װ�ã��������ռ���������������
| Ũ���� |
| ���� |
�ʴ�Ϊ��Ӧ�ȼ����Ҵ���Ȼ���ҡ���Թܱ���������Ũ���ᣬ����������CH3COOH+CH3CH2OH
| Ũ���� |
| ���� |
��2������������ԣ��ܺͱ���̼������Һ��Ӧ���ѱ����գ����������ڱ���̼������Һ�е��ܽ�Ƚ�С�����ڷ��룬
�ʴ�Ϊ��BC����
��3����Ӧ�����Ҵ�������ķе�ϵͣ����ô����ȣ�������Ӧ���������������ʧԭ�ϣ��¶ȹ������ܷ�����������Ӧ������Ϊ��ֹ�Ҵ�������ӷ������ԭ�ϵ���ʧ��ӦС����ȣ�
�ʴ�Ϊ�����������Ҵ��Ļӷ������ٸ���Ӧ�ķ�����
��4��̼����ˮ��ʼ��ԣ��������������ڱ���̼������Һ���ܶȱ�ˮС������ζ����ʱ�����̼���Ʒ�Ӧ��ʹ��Һ��ɫ��dz��������������������ˮ�����Ƴ�ȥ������ˮ����ˮ��������ˮ�γ������ƽᾧˮ�������ѡ��P2O5����ʯ�Һ�NaOH�ȹ����������Է��������������ԣ�P2O5��ˮ�����ᣩ�����������ˮ�⣬
�ʴ�Ϊ���Թ�B�е�Һ��ֳ��������㣬�ϲ���ɫ���²�Ϊ��ɫҺ�壬���²�Һ��ĺ�ɫ��dz��B����
��5���Ӹ�װ���봫ͳ�Ʊ�װ�õ�ʹ�õ������IJ�ͬ���з������������¶ȼƣ������ڿ��Ʒ���װ���з�ӦҺ���¶ȣ����ݷ�Ӧ�Ŀ������ص㣬���ӷ�Ӧ��������ƽ�����������ƶ���������������IJ���������������װ�ã��������ռ����
�ʴ�Ϊ�����������¶ȼƣ����ڿ��Ʒ���װ���з�ӦҺ���¶ȣ����ٸ�����ķ������������˷�Һ©���������ڼ�ʱ���䷴Ӧ���Һ����������������IJ�����������������װ�ã��������ռ���������������
������������Ҫ�����������������Ʊ������������������Ʊ�ԭ�����Լ�����ԭ������ʵ��װ�õĸĽ�����˼·�ǽ��Ĺؼ�����Ŀ�Ѷ��еȣ�

��ϰ��ϵ�д�
 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�
�����Ŀ
��֪�������ݣ�
2Fe��s��+O2��g��=2FeO��s����H=-544kJ?mol-1
4Al��s��+3O2��g��=2Al2O3��s����H=-3350kJ?mol-1
��2Al��s��+3FeO��s��=Al2O3��s��+3Fe��s���ġ�H�ǣ�������
2Fe��s��+O2��g��=2FeO��s����H=-544kJ?mol-1
4Al��s��+3O2��g��=2Al2O3��s����H=-3350kJ?mol-1
��2Al��s��+3FeO��s��=Al2O3��s��+3Fe��s���ġ�H�ǣ�������
| A��-859 kJ?mol-1 | B��+859 kJ?mol-1 | C��-1403 kJ?mol-1 | D��-2491 kJ?mol-1 |
 ij��ѧ��Ӧ�У��跴Ӧ���������ΪE1���������������ΪE2��
ij��ѧ��Ӧ�У��跴Ӧ���������ΪE1���������������ΪE2��