��Ŀ����
�����ӵ�����ԼΪ6.02��1023mol��1������������ȷ����
| A��4 g��ˮ��D2O��������������Ϊ0.2��6.02��1023 |
| B��2.8 g��ϩ�ͱ�ϩ�Ļ������������̼ԭ����Ϊ0.2��6.02��1023 |
| C��4.48 L H2��O2�Ļ������������������Ϊ0.2��6.02��1023 |
| D��0.2 mol Cl2�ܽ��ڵ������ˮ�У�ת�Ƶ�����Ϊ0.2��6.02��1023 |
A
A��4 g��ˮ��D2O��������������Ϊ������0.2��6.02��1023������0.2 mol Cl2�ܽ��ڵ������ˮ�У�ת�Ƶ�����С��0.2��6.02��1023����ΪCl2��ˮ�ķ�Ӧ�ǿ���ġ��ºͣÿ���ȥ���ƣ�������Ħ�����������¶ȡ�ѹǿ�仯���仯��������Ħ���������֮�仯������δ˵���Ƿ��״���������£���ȷ���ô���

��ϰ��ϵ�д�
 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�
�����Ŀ
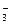 ����
���� ������ΪNA
������ΪNA
 00mL����ƿ�У��ձ�δϴ�ӡ�
00mL����ƿ�У��ձ�δϴ�ӡ�