��Ŀ����
�����������������õķ���ʴ�ԣ��ڹ�����ҵ���зdz���Ҫ�����á���������пհף�
(1)��19����ʱ������һ�����Ľ�������������õ���������ͬ�䱦�����ļ۸�ͬ�ƽ��൱��1827�꣬�¹���ѧ��ά���ý���������ˮ�Ȼ�����Ӧ���Ƶ��˽��������ü�����ˮ�Ȼ�����Ӧ�����������Ȼ�����Һ��������_________�� �ִ���ҵ������ԭ��������������ȡ���ã�����ȡ������ͨ�������Ϊ____��
(2)����͵�������ı������������ʡ���ʯī�����ۺͶ������Ѱ�һ����������ڸ��������գ��������ʿ������²��ϣ�4Al(s)+3TiO2��s��+3C(s)  2Al2O3(s)+3TiC(s)
2Al2O3(s)+3TiC(s)
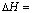 -1176 kJ��mol-1����Ӧ�����У�ÿת��1 mol���ӷų�������Ϊ________��
-1176 kJ��mol-1����Ӧ�����У�ÿת��1 mol���ӷų�������Ϊ________��
(3)������ͬ���ڣ��ؿ�������ĺ����ǹ�________������>��<��=���� Na2CaSi6O14�ǹ����β�������Ҫ�ɷ֣�Na2CaSi6O14Ҳ��д��Na2O��CaO��6SiO2��ʢ��NaOH��Һ���Լ�ƿ���ò���ƿ�������γ�ճ�ԵĹ����ζ�����������Ӧ�Ļ�ѧ����ʽ ________����ʯ���������ᣬ��ͬ�ʯ����ԭ�ӵ����ʵ���������ͬ�����Ƴ�ʯ��ѧʽNaAlSi3O8����֪�Ƴ�ʯ�Ļ�ѧʽΪ________��
|
 3Ba��+BaO��Al2O3, �û�ѧƽ���ƶ�ԭ������������������ȡ����Ba��ԭ����_______________________________.
3Ba��+BaO��Al2O3, �û�ѧƽ���ƶ�ԭ������������������ȡ����Ba��ԭ����_______________________________.
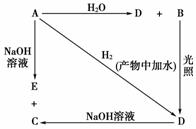
��1��������ˮ��Ӧ����KOH��KOH�����Ȼ�����Ӧ�����������������õ���������2�֣��� CO2��2�֣�
��2��98 kJ��2�֣�
��3������2�֣��� SiO2��2NaOH��Na2SiO3��H2O��2�֣��� CaAl2Si2O8 ��2�֣�
��4������Ba�ķе��Al�ĵͣ�Ba�������ݳ���ʹƽ�����ơ���2�֣�

 2Z���ﵽƽ���û�������з�Ӧ��������ʵ�����������������ʵ���֮��Ϊ3:2����Y��ת������ӽ���( )
2Z���ﵽƽ���û�������з�Ӧ��������ʵ�����������������ʵ���֮��Ϊ3:2����Y��ת������ӽ���( )