��Ŀ����
��2012?����һģ��ʵ������NH4HCO3��NaHSO3�ĸ����״����ij��ȤС��Ϊ�ⶨ����NH4HCO3�ĺ�������������ڻ�����м����ʵ�鷽�����ⶨ�������������ش��������⣺
��1����ѧС����Ҫѡ��������ҩƷ����������ʵ�飮��ͼ��ÿ������װ��ֻѡ����һ�Σ�����̨�ȹ̶�����δ������
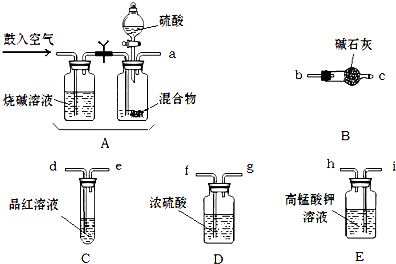
�밴�����������ҵķ�����������˳���ǣ��������Ľӿ���ĸa��b����
a��
��2��ʵ�鿪ʼ����������ǰ��Aװ����Ҫ���������������
��3��Eװ�õ�������
��4��ʵ���У���Cƿ����Һ��ɫ����ⶨ������ܻ�ƫ
mL��
��5�����������Ʒ����Ϊ13.1g��������ú��ʯ������4.4g����������NH4HCO3����������Ϊ
��6���Ӷ����ⶨ��ȷ�Կ��ǣ�����װ�û�Ӧ����һ���ĸĽ���
�����õ������ԭ��������H��1 C��12 N��14 O��16 Na��23 S��32��
��1����ѧС����Ҫѡ��������ҩƷ����������ʵ�飮��ͼ��ÿ������װ��ֻѡ����һ�Σ�����̨�ȹ̶�����δ������

�밴�����������ҵķ�����������˳���ǣ��������Ľӿ���ĸa��b����
a��
i
i
��h
h
��d
d
��e
e
��f��g��b
b
��c
c
����2��ʵ�鿪ʼ����������ǰ��Aװ����Ҫ���������������
�ų�װ����ԭ�п���
�ų�װ����ԭ�п���
�������Aװ���ٴι�������������������ɵ�����ȫ���������װ��
�����ɵ�����ȫ���������װ��
����3��Eװ�õ�������
���ն����������壬��ֹ��������Բⶨ�����Ӱ��
���ն����������壬��ֹ��������Բⶨ�����Ӱ��
����4��ʵ���У���Cƿ����Һ��ɫ����ⶨ������ܻ�ƫ
��
��
����ߡ��͡�����Ϊ��ȷ��ʵ����Cƿ��Һ����ɫ����ȡ��Ʒ������Ϊm g��ʵ��ǰ E����װ a mol?L-1��KMnO4��Һ�����V��������| 50m |
| 13a |
| 50m |
| 13a |
��5�����������Ʒ����Ϊ13.1g��������ú��ʯ������4.4g����������NH4HCO3����������Ϊ
60.3%
60.3%
����6���Ӷ����ⶨ��ȷ�Կ��ǣ�����װ�û�Ӧ����һ���ĸĽ���
��B֮���ٽ�һ����ֹ������ˮ�Ͷ�����̼����B��װ��
��B֮���ٽ�һ����ֹ������ˮ�Ͷ�����̼����B��װ��
�������õ������ԭ��������H��1 C��12 N��14 O��16 Na��23 S��32��
��������1������ʵ��ԭ�����������м������������̼�Ͷ����������壬�ø��������Һ��ȥ��������Ȼ��ͨ��Ʒ����Һ������������Ƿ���������Ž�����ͨ��Ũ����������ü�ʯ�����ն�����̼���壬��ʯ�����ӵ�������Ϊ������̼�����������
��2��ʵ�鿪ʼ����������ǰ��Aװ����Ҫ�������Ŀ�����ų�ϵͳ�еĶ�����̼���壬������ٴι��������������ʹ���ɵ�����ȫ���������װ�ã�
��3�����������Һ�����������ն����������壻
��4����Ʒ����Һ��ɫ��˵��������������û�г�����������ʯ�����գ����Լ�ʯ��������ƫ���ٸ���ʵ��ԭ�����������ݶ�������������֮��Ĺ�ϵʽ���㣻
��5����ʯ������4.4g��������̼���ʵ���Ϊ0.1mol����̼Ԫ���غ㣬̼��������ʵ���ҲΪ0.1mol��̼���������Ϊ7.9g��������NH4HCO3������������
��6����ʯ�������տ����еĶ�����̼��ˮ������
��2��ʵ�鿪ʼ����������ǰ��Aװ����Ҫ�������Ŀ�����ų�ϵͳ�еĶ�����̼���壬������ٴι��������������ʹ���ɵ�����ȫ���������װ�ã�
��3�����������Һ�����������ն����������壻
��4����Ʒ����Һ��ɫ��˵��������������û�г�����������ʯ�����գ����Լ�ʯ��������ƫ���ٸ���ʵ��ԭ�����������ݶ�������������֮��Ĺ�ϵʽ���㣻
��5����ʯ������4.4g��������̼���ʵ���Ϊ0.1mol����̼Ԫ���غ㣬̼��������ʵ���ҲΪ0.1mol��̼���������Ϊ7.9g��������NH4HCO3������������
��6����ʯ�������տ����еĶ�����̼��ˮ������
����⣺��1������ʵ��ԭ�����������м������������̼�Ͷ����������壬�ø��������Һ��ȥ��������Ȼ��ͨ��Ʒ����Һ������������Ƿ���������Ž�����ͨ��Ũ����������ü�ʯ�����ն�����̼���壬��ʯ�����ӵ�������Ϊ������̼���������������װ������˳����aihdefgbc���ʴ�Ϊ��i h d e b c��
��2��ʵ�鿪ʼ����������ǰ��Aװ����Ҫ�������Ŀ�����ų�ϵͳ�еĶ�����̼���壬������ٴι��������������ʹ���ɵ�����ȫ���������װ�ã�
�ʴ�Ϊ���ų�װ����ԭ�п����������ɵ�����ȫ���������װ�ã�
��3�����������Һ�����������ն����������壬��ֹ��������Բⶨ�����Ӱ�죬�ʴ�Ϊ�����ն����������壬��ֹ��������Բⶨ�����Ӱ�죻
��4����Ʒ����Һ��ɫ��˵��������������û�г�����������ʯ�����գ����Լ�ʯ��������ƫ����ʵ��ԭ��������֪������̼���������ƫ����������NH4HCO3ƫ�ⶨ�����ƫ�ߣ�
���������Һ��ǿ�����ԣ��ܽ�������������������������ӣ���Ӧ����ʽΪ��5SO2+2KMnO4+2H2O=K2SO4+2MnSO4+2H2SO4��������Ʒȫ�������������ƣ�������ԭ���غ�ö�����������ʵ���=
mol��
���ݶ�������������֮��Ĺ�ϵʽ�ã�������ص����=
=
mL��
�ʴ�Ϊ���ߣ�
��
��5����ʯ������4.4g����������̼���ʵ���Ϊ0.1mol����̼Ԫ���غ㣬̼��������ʵ���ҲΪ0.1mol��̼���������Ϊ7.9g��NH4HCO3����������Ϊ
%=60.3%���ʴ�Ϊ��60.3%��
��6����ʯ���������ͨ����ʯ�������տ����еĶ�����̼��ˮ����������Ӧ�ڼ�ʯ�Һ���������һ��װ�м�ʯ�ҵ�װ�ã�
�ʴ�Ϊ����B֮���ٽ�һ����ֹ������ˮ�Ͷ�����̼����B��װ�ã�
��2��ʵ�鿪ʼ����������ǰ��Aװ����Ҫ�������Ŀ�����ų�ϵͳ�еĶ�����̼���壬������ٴι��������������ʹ���ɵ�����ȫ���������װ�ã�
�ʴ�Ϊ���ų�װ����ԭ�п����������ɵ�����ȫ���������װ�ã�
��3�����������Һ�����������ն����������壬��ֹ��������Բⶨ�����Ӱ�죬�ʴ�Ϊ�����ն����������壬��ֹ��������Բⶨ�����Ӱ�죻
��4����Ʒ����Һ��ɫ��˵��������������û�г�����������ʯ�����գ����Լ�ʯ��������ƫ����ʵ��ԭ��������֪������̼���������ƫ����������NH4HCO3ƫ�ⶨ�����ƫ�ߣ�
���������Һ��ǿ�����ԣ��ܽ�������������������������ӣ���Ӧ����ʽΪ��5SO2+2KMnO4+2H2O=K2SO4+2MnSO4+2H2SO4��������Ʒȫ�������������ƣ�������ԭ���غ�ö�����������ʵ���=
| m |
| 104 |
���ݶ�������������֮��Ĺ�ϵʽ�ã�������ص����=
| ||||
| amol/L |
| 50m |
| 13a |
�ʴ�Ϊ���ߣ�
| 50m |
| 13a |
��5����ʯ������4.4g����������̼���ʵ���Ϊ0.1mol����̼Ԫ���غ㣬̼��������ʵ���ҲΪ0.1mol��̼���������Ϊ7.9g��NH4HCO3����������Ϊ
| 7.9g |
| 13.1g |
��6����ʯ���������ͨ����ʯ�������տ����еĶ�����̼��ˮ����������Ӧ�ڼ�ʯ�Һ���������һ��װ�м�ʯ�ҵ�װ�ã�
�ʴ�Ϊ����B֮���ٽ�һ����ֹ������ˮ�Ͷ�����̼����B��װ�ã�
������������ҪĿ���Dzⶨ������̼���������������������Ҳ�ܱ���ʯ�����գ����������ն�����̼����ǰӦ���������������������ȥ������������һ�������Ը��������Һ����ˮ������Ʒ����Һ����������������Ƿ������

��ϰ��ϵ�д�
�����Ŀ
��2012?����һģ������ʵ�������Ԥ��ʵ��Ŀ�Ļ�����ʵ�����һ�µ��ǣ�������
|