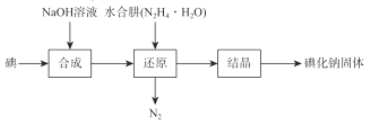
��Ŀ����
����Ŀ��������������Ҫ������������ԭ��������ˮ��Һ�ֳ�Ϊ˫��ˮ��������������ɱ����Ư�ȡ�ij��ѧ��ȤС��ȡһ�����Ĺ���������Һ��ȷ�ⶨ�˹�������ĺ���������д���пհף�
��ȡ10.00 mL�ܶ�Ϊ�� g/mL�Ĺ�������ԭ��Һϡ�ͳ�250 mL����ȡϡ����������Һ25.00 mL����ƿ�У�����ϡ�����ữ��������ˮϡ�ͣ�������������
��1���ø�����ط�(һ��������ԭ�ζ���)�ɲⶨ����Һ�е�H2O2�ĺ�������������Ũ��Ϊ0.10 mol��L1��KMnO4����Һ500 mL��Ӧȷ��ȡ_________g KMnO4[��֪M(KMnO4)=158.0 g��mol1]��
a.���Ƹñ���Һʱ�����������в���Ҫ�õ�����_________��(�ñ�ű�ʾ)��
��������ƽ ���ձ� ����Ͳ �������� ������ƿ ����ͷ�ι� ����Һ��
b.����ʱ���Ӷ������������ս��__________(�ƫ��ƫС�����䡱)��
��2����ɲ���ƽ���ӷ���ʽ��
![]()
![]() ��
��![]() H2O2��
H2O2��![]()
![]()
![]() Mn2+��
Mn2+��![]() O2����
O2����![]()
��3���ζ�ʱ����������ر���Һע��____________(���ʽ����ʽ��)�ζ����С��ζ������յ��������____________________________________________��
��4���ظ��ζ����Σ�ƽ������KMnO4����ҺV mL����ԭ����������Һ�й����������������Ϊ_____________��
��5�����ζ�ǰ�ζ��ܼ����������ݣ��ζ���������ʧ����ⶨ���______________ (�ƫ�ߡ���ƫ�͡����䡱)��
���𰸡���1��7.9 ���� ƫС
��2��2 5 6 H+ 2 5 8H2O
��3����ʽ�������һ�θ��������Һ����Һ����ɫ��Ϊdz��ɫ(����ɫ)����30���ڲ���ɫ
��4��![]()
��5��ƫ��
�����������⿼����һ�����ʵ���Ũ����Һ�����Ƽ����������ζ���������ԭ��Ӧ����ƽ����1���ø�����ط�(һ��������ԭ�ζ���)�ɲⶨ����Һ�е�H2O2�ĺ�������������Ũ��Ϊ0.10 mol��L1��KMnO4����Һ500 mL��Ӧȷ��ȡ������ص�����Ϊ��0.1 mol/L��0.5 L��158.0 g/mol=7.9 g���ù���������Һ��Ҫ��������ƽ�������������������ձ����ܽ⣬Ȼ��ת�Ƶ�����ƿ�У�����Ҫ��ȡҺ�����Ͳ����Һ�ܣ�����ʱ���Ӷ�������ʹ������Һ���������Ũ�ȼ�С��
��2�����������Mn�Ļ��ϼ۴ӣ�7�۽��ͣ�2�ۣ�˫��ˮ�е���Ԫ�ش�1�����ߵ�0�ۣ��������������������Ը��ݵ��ӵ�ʧ�غ��֪���������ͻ�ԭ�������ʵ���֮����2��5���ٸ��������غ�͵���غ�ɵû�ѧ����ʽ��2![]() ��5H2O2��6H+
��5H2O2��6H+![]() 2Mn2+��5O2����8H2O��
2Mn2+��5O2����8H2O��
��3�����������Һ���������ԣ��ܸ�ʴ������Ӧ������ʽ�ζ��ܡ��������Ը��������Һ�����Ϻ�ɫ�ģ������յ�������ǵ���һ�θ��������Һ����Һ��dz��ɫ����30���ڲ���ɫ��
��4�����ݻ�ѧ����ʽ���Եõ���ϵʽ��2![]() ~5H2O2������0.1 mol/L KMnO4����ҺV mL��������0.1V��103 mol�ĸ������ʱ������˫��ˮ�����ʵ�����0.25V��103 mol����ԭ�������������Ϊ��0.00025V mol��34 g/mol=0.0085V g������������Һ�й����������������Ϊ��
~5H2O2������0.1 mol/L KMnO4����ҺV mL��������0.1V��103 mol�ĸ������ʱ������˫��ˮ�����ʵ�����0.25V��103 mol����ԭ�������������Ϊ��0.00025V mol��34 g/mol=0.0085V g������������Һ�й����������������Ϊ��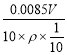 =
=![]() ��
��
��5�����ζ�ǰ�ζ��ܼ����������ݣ��ζ���������ʧ�������������Ը��������Һ���ƫ�����Բⶨ���ƫ�ߡ�

����Ŀ����17�֣��±���Ԫ�����ڱ���һ����, ��Ա��е��١�����Ԫ��,��д���пհ�(��д��Ų��÷�):
���� ���� | ��A | ��A | ��A | ��A | ��A | ��A | ��A | 0�� |
2 | �� | �� | �� | |||||
3 | �� | �� | �� | �� | �� | |||
4 | �� | �� |
��1������ЩԪ����,��ѧ��������õ���:_____������Ԫ�ط�����
��2��������������ˮ�����У�������ǿ�Ļ��������ѧʽ��/span>______��������ǿ�Ļ��������ѧʽ��:__________��
��3���ȽϢ���ݵ�����������Ӧ��ˮ���_________������ǿ������ѧʽ������ͨ��________________________________˵����д��Ӧ����ѧ����ʽ����
��4��ʵ������ȡ�ڵ��⻯�����ѧ����ʽ_______________________________ ���ڵ��⻯����ڵ�����������ˮ���ﷴӦ���õIJ�����ѧʽΪ______
��5���������γɶ������������һ���Ǻ���ɫ���壬���÷���ʽ˵�������岻�˲�����ˮ���ռ���ԭ��________________������ѧ����ʽ��ʾ��
��6���ȽϢ�����⻯�_________ ���ȶ�������ѧʽ��
��7��д���ܵĵ�����ˮ��Ӧ�����ӷ���ʽ_______________________��
��8��д����Ԫ�ص������ṹʾ��ͼ______,�����Ӱ뾶_________ S2-�������������д����Ԫ�������ڱ���λ��_______________________