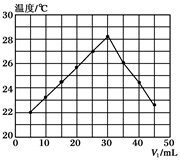
��Ŀ����
��֪BrCl�ܷ������з�Ӧ��MΪ+n�۵Ľ���Ԫ�أ���
nBrCl + 2M ="==" MCl n + MBr n BrCl + H2O ="==" HCl + HBrO
���½��۲���ȷ���ǣ�
nBrCl + 2M ="==" MCl n + MBr n BrCl + H2O ="==" HCl + HBrO
���½��۲���ȷ���ǣ�
| A��BrCl���к�±�ص������ƵĻ�ѧ���� |
| B��BrCl��M��Ӧʱ��BrClֻ�������� |
| C��BrCl��NaOH��Һ��Ӧ����NaCl��NaBrO������ |
| D��BrCl��H2O�ķ�Ӧ�У�BrCl����������������ԭ�� |
D
���������BrCl������±�أ����к�±�ص������ƵĻ�ѧ���ʣ���A��ȷ��
����MΪ+n�۵Ľ���Ԫ�أ�����ԭ��������BrCl������Ӧʱ��BrClֻ��������������B��ȷ��
BrCl��NaOH��Һ��Ӧ������BrCl������Cl2,��Ӧ����NaCl��NaBrO�����Σ���C��ȷ��
D��BrCl + H2O ="==" HCl + HBrO��û�з������ϼ۵ı仯����D����
�������������һ�����Ѷȣ���Ҫ����±�صĻ�ѧ���ʣ�������ԭ��Ӧ��ֻҪ�������֪ʶ���ɽ��

��ϰ��ϵ�д�
 �����������һ��һ��ϵ�д�
�����������һ��һ��ϵ�д�
�����Ŀ
 ��NH4Cl�ĵ���ʽ��
��NH4Cl�ĵ���ʽ��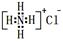
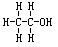 ��Na�Ľṹʾ��ͼ��
��Na�Ľṹʾ��ͼ�� ������ķ���ʽ�� CH3COOH
������ķ���ʽ�� CH3COOH 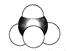 ������������ϩ�����ģ�ͣ�
������������ϩ�����ģ�ͣ�