��Ŀ����
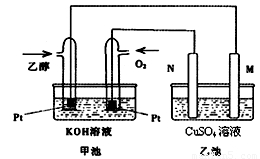
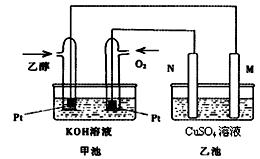
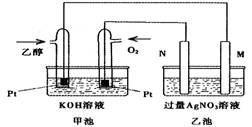
��ͼ��һ���Ҵ�ȼ�ϵ�ع���ʱ��ʾ��ͼ���ҳ��е������缫һ����ʯī�缫��һ�������缫������ʱM��N�����缫�������������٣���ش��������⣺
��1��M�缫�IJ������������� ���缫�������������� ��
�����Ҵ��IJ��缫�ĵ缫��ӦʽΪ���������������� ������������ ��
д���ҳ��з����Ļ�ѧ��Ӧ�����ӷ���ʽ������������������������������ ��
��2���ڴ˹����У��ҳ���ijһ�缫����������4.32gʱ���׳�����������������Ϊ���� L����״���£�������ʱ�ҳ���Һ�����Ϊ400mL�����ҳ�����Һ��pHΪ�� ��
��3�����ڳ��³�ѹ�£�1g C2H5OHȼ������CO2��Һ̬H2Oʱ�ų�29.71kJ������
ʾ�÷�Ӧ���Ȼ�ѧ����ʽΪ������������������������������������������ ��
��4������Ҳ��һ�ֺܺõ������Դ���̲��ں��ġ���ȼ�����Ǹ�ѹ���γɵ��������ļ���ˮ������塣��������ȼ�յ��Ȼ�ѧ����ʽΪ��
CH4(g)��2O2(g)��CO2(g)��2H2O(l) ��H����890.3 kJ��mol��
356g����ȼ����(������ʽΪCH4��9H2O)�ͷŵļ���������ȫȼ������Һ̬ˮ���ų�������Ϊ�������������� ��kJ��
��1������1�֣� ���� ��1�֣�4OH����4e�� 2H2O+O2����2�֣�
C2H5OH��12e+16OH��=2CO32��+11H2O��2�֣�
��2��0.224��2�֣� 1��2�֣�
��3��C2H5OH��l��+3O2��g�� 2CO2��g��+3H2O��l�� ��H=��1366.7kJ/mol ��2�֣�
��4��1780.6��2�֣�

 ��)����������ͨ���Ҵ� g��
��)����������ͨ���Ҵ� g��
 A.M�缫�IJ�����ʯī
A.M�缫�IJ�����ʯī