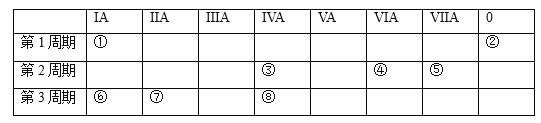
题目内容
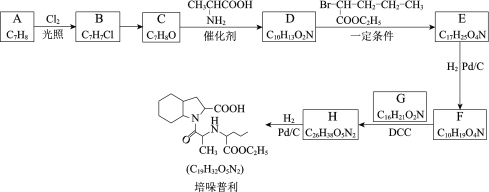
【题目】培哚普利主要用于治疗高血压与充血性心力衰竭,其合成路线如下:
已知:ⅰ. ![]()
ⅱ.DCC是一种很强的脱水剂。
(1)芳香烃A的名称是___________________。
(2)B→C所需的试剂及条件是________________。
(3)C→D的化学方程式是____________________。
(4)D→E的反应类型是______________________。
(5)F的结构简式是____________________________。
(6)K存在互变异构体K′,G可由如下路线合成:
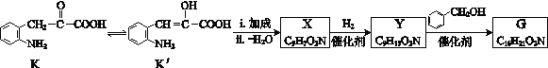
①X含有五元环和碳碳双键,其结构简式是______。
②上述合成培哚普利的路线中,将Y转化为G,再与F反应,而不直接用Y的主要目的是______,与合成路线中______________填字母)的转化目的相同。
【答案】甲苯 NaOH的水溶液加热 ![]() 取代反应
取代反应 ![]()
![]() 保护Y中的羧基 C→D
保护Y中的羧基 C→D
【解析】
结合逆合成分析法,H在Pd/C作用下与H2发生已知信息ⅰ的反应,根据H和培哚普利的分子式,可得H的结构简式为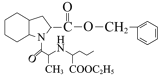 ,DCC是一种很强的脱水剂,F和G发生脱水反应生成H,结合G、F的分子式和H的结构式,可推知F的结构简式为
,DCC是一种很强的脱水剂,F和G发生脱水反应生成H,结合G、F的分子式和H的结构式,可推知F的结构简式为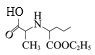 ,G的结构简式为
,G的结构简式为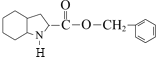 ,E在Pd/C作用下与H2发生已知信息ⅰ的反应,根据F的结构式即分子式和E的分子式,E的结构简式为
,E在Pd/C作用下与H2发生已知信息ⅰ的反应,根据F的结构式即分子式和E的分子式,E的结构简式为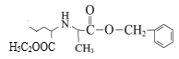 ,结合D和E的分子式和E结构式,D和
,结合D和E的分子式和E结构式,D和![]() 在一定条件下发生取代反应生成E,D的结构简式为
在一定条件下发生取代反应生成E,D的结构简式为 ,C与
,C与![]() 在催化剂的作用下生成D,结合分子式,C的结构简式为
在催化剂的作用下生成D,结合分子式,C的结构简式为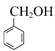 ,B在一定条件下发生卤代烃的水解生成C,则B的结构简式为
,B在一定条件下发生卤代烃的水解生成C,则B的结构简式为![]() ,A在光照的条件下与Cl2发生取代反应生成B,A的结构简式为
,A在光照的条件下与Cl2发生取代反应生成B,A的结构简式为![]() ,据此分析解答。
,据此分析解答。
(1)根据分析芳香烃A的结构简式为![]() ,名称是甲苯;
,名称是甲苯;
(2)根据分析,B在一定条件下发生卤代烃的水解生成C,则B的结构简式为![]() ,卤代烃发生水解反应的条件为NaOH的水溶液加热;
,卤代烃发生水解反应的条件为NaOH的水溶液加热;
(3) D的结构简式为 ,C与
,C与![]() 在催化剂的作用下生成D,结合分子式,C的结构简式为
在催化剂的作用下生成D,结合分子式,C的结构简式为 ,化学方程式是
,化学方程式是 +
+![]()
![]()
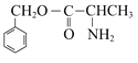 +H2O;
+H2O;
(4)根据分析,D→E的反应类型是取代反应;
(5)F的结构简式是![]() ;
;
(6)①K′发生加成反应,在发生分子内脱水,结合X的分子式,以及X含有五元环和碳碳双键,其结构简式是![]() ;
;
②上述合成培哚普利的路线中,将Y转化为G,再与F反应,而不直接用Y的主要目的是保护Y中的羧基,与合成路线中C→D的转化目的相同。

 阅读快车系列答案
阅读快车系列答案【题目】一定条件下,通过下列反应可以制备特种陶瓷的原料MgO, MgSO3(s) + CO(g)![]() MgO(s) + CO2(g) +SO2(g) △H>0。该反应在恒容的密闭容器中达到平衡后,若仅改变图中横坐标x的值,重新达到平衡后,纵坐标y随x变化趋势合理的是
MgO(s) + CO2(g) +SO2(g) △H>0。该反应在恒容的密闭容器中达到平衡后,若仅改变图中横坐标x的值,重新达到平衡后,纵坐标y随x变化趋势合理的是

选项 | x | y |
A | 温度 | 容器内混合气体的密度 |
B | CO的物质的量 | CO2与CO的物质的量之比 |
C | SO2的浓度 | 平衡常数K |
D | MgSO4的质量(忽略体积) | CO的转化率 |