��Ŀ����
ʵ���ҿ������̿�(��Ҫ�ɷ�ΪMnO2�Ʊ�KMnO4���������£����̿����������KOH��KClO3�ڸ����·�Ӧ�����������(K2MnO4)��KCl����ˮ�ܽ⣬��ȥ��������Һ�ữ��K2MnO4ת��ΪMnO2��KMnO4����ȥMnO2������Ũ����Һ���ᾧ�õ�����ɫ����״KMnO4�� ��ش�
(1)���̿��Ʊ�K2MnO4�Ļ�ѧ����ʽ��__________________ ��
(2)K2MnO4�Ʊ�KMnO4�����ӷ���ʽ��_____________________��
(3)����2��5g���̿�(��MnO280%)��������ʵ�飬����KMnO4�����۲�����
(4)KMnO4�����ȵľ������ữ��Na2C2O4��Ӧ����Mn2+��CO2���÷�Ӧ�Ļ�ѧ����ʽ��________________________��
(5)�����Ƶõ�KMnO4��Ʒ0��165g��ǡ����0��335g��Na2C2O4��Ӧ��ȫ�������KMnO4�Ĵ��ȡ�
(1)���̿��Ʊ�K2MnO4�Ļ�ѧ����ʽ��__________________ ��
(2)K2MnO4�Ʊ�KMnO4�����ӷ���ʽ��_____________________��
(3)����2��5g���̿�(��MnO280%)��������ʵ�飬����KMnO4�����۲�����
(4)KMnO4�����ȵľ������ữ��Na2C2O4��Ӧ����Mn2+��CO2���÷�Ӧ�Ļ�ѧ����ʽ��________________________��
(5)�����Ƶõ�KMnO4��Ʒ0��165g��ǡ����0��335g��Na2C2O4��Ӧ��ȫ�������KMnO4�Ĵ��ȡ�
(1)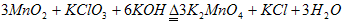
(2)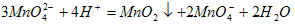
(3)����(1)��(2)�е���������ʽ���Եõ���ϵʽ��
3MnO2��������2KMnO4
3��87 2��158
2.5g��80�� m(KMnO4)
m(KMnO4)=2��158��2��5g��80����(3��87)=2��4g
(4)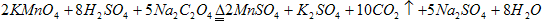
(5)����(4)�еĻ�ѧ����ʽ��
2KMnO4����������5Na2C2O4
2��158 5��134
m(KMnO4) 0.335g
m(KMnO4)=2��158��0��335g��(5��134)=0��158g
KMnO4����=(0��158g��0��165g)��100��=95��8��
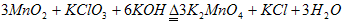
(2)
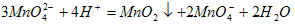
(3)����(1)��(2)�е���������ʽ���Եõ���ϵʽ��
3MnO2��������2KMnO4
3��87 2��158
2.5g��80�� m(KMnO4)
m(KMnO4)=2��158��2��5g��80����(3��87)=2��4g
(4)
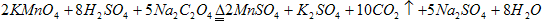
(5)����(4)�еĻ�ѧ����ʽ��
2KMnO4����������5Na2C2O4
2��158 5��134
m(KMnO4) 0.335g
m(KMnO4)=2��158��0��335g��(5��134)=0��158g
KMnO4����=(0��158g��0��165g)��100��=95��8��

��ϰ��ϵ�д�
�����Ŀ