��Ŀ����
����12�֣�
��1����CO2Ϊ̼Դ��ȡ��̼�л���һֱ�ǻ�ѧ������о��ȵ㣬CO2������ȡ��̼���ķ�Ӧ���£�
��ӦI��CO2(g)+3H2(g)=CH3OH(g)+H2O(g) ��H=��49.0kJ/mol
��ӦII��2CO2(g)+6H2(g)=CH3CH2OH(g)+3H2O(g) ��H=��173.6kJ/mol
д����CH3OH(g)�ϳ�CH3CH2OH(g)�ķ�Ӧ���Ȼ�ѧ����ʽ_______________
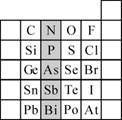
��2��������أ�K2FeO4��������һ����Ҫ��������м�ǿ��������
�ٵ�ⷨ�ǹ�ҵ���Ʊ�K2FeO4��һ�ַ���������Ϊ�����������������Һ��Ȼ����������Һ�м���KOH�����ʱ����������Ӧ����FeO42-���õ缫��ӦʽΪ_________________
����MnO2��Zn������ƣ�K2FeO4��ZnҲ������ɼ��Ե�أ�K2FeO4�ڵ�������������ϣ���缫��ӦʽΪFeO42-+3e-+4H2O=Fe(OH)3+5OH-����õ���ܷ�Ӧ�����ӷ���ʽΪ__________________
��3��amol FeS��bmol FeOͶ�뵽VL��C mol/L��������Һ�г�ַ�Ӧ����NO���壬���ó�����Һ�ɷֿɿ�����Fe(NO3)3��H2SO4�Ļ��Һ����Ӧ��δ����ԭ���������Ϊ_____
�٣�a+b����63g �ڣ�a+b����189g �ۣ�a+b��mol ��VC��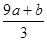 mol
mol
��1��2CH3OH(g)=CH3CH2OH(g)+H2O(g) ��H= -75.6kJ/mol
��2����Fe��6e- +8OH- = FeO42- + 4H2O
��3Zn + 2FeO42- + 8H2O = 2Fe(OH) 3 + 3Zn(OH)2 + 4OH-
��3���ڢ�
��������
�����������1�����ݸ�˹���ɿ�֪����-2���ٵõ�Ŀ�귴Ӧ��������CH3OH(g)�ϳ�CH3CH2OH(g)���Ȼ�ѧ����ʽΪ��2CH3OH(g)=CH3CH2OH(g)+H2O(g) ��H= -75.6kJ/mol��
��2������������������ʧ���ӣ����Ե缫��ӦΪFe��6e- +8OH- = FeO42- + 4H2O
��ZnΪ���������Ը����ĵ缫��ӦΪZn��2e-+2OH-= Zn(OH)2�������ܵ����ӷ���ʽΪ3Zn + 2FeO42- + 8H2O = 2Fe(OH) 3 + 3Zn(OH)2 + 4OH-��
��δ����Ӧ�����������Һ�е�NO3-����Ϊ��Ӧ�����Һ���Կ�����Fe(NO3)3��H2SO4�Ļ��Һ��������Һ�е�NO3-���Ը���Fe3+���������㣬��3(a+b)mol��63g/mol=(a+b)��189g�������Ը�������������㣬������������Ӧ��һ������������ת��ΪNO��һ����δ����ԭ��NO3-����ʽ��������Һ�У����ݵ�ʧ��������ȿ����n(NO)=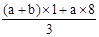 =
=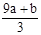 ������NO3-����ΪVC-
������NO3-����ΪVC-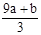 ��
��
���㣺��ѧ��Ӧ�е������仯��ԭ��ء����ء�����ļ���
�����������ۺ���ǿ���ѶȽϴ���Ҫ����ѧ��������������������ڸ߿��ȵ�ϰ�⡣

 ��ɫ
��ɫ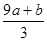 mol
mol