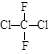��Ŀ����
(10��)д�����и��л���Ľṹ��ʽ��
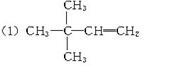
��1����0.2 mol��A����������ȫȼ��ʱ����CO2��H2O��1.2 mol�������������2��2-�������飬��A�ĽṹʽΪ___________��
��2������ʽΪC6H12��ijϩ�������е�̼ԭ�Ӷ���ͬһƽ���ϣ����ϩ���Ľṹ��ʽΪ______________________��
��3��ij����A�����ܶ�����ͬ״���������ܶȵ�64�������ⶨ֪A�����й���6������
����A��������ϩ���������ӳɵIJ��A�Ľṹ��ʽΪ______________________��
����A��Ȳ���������ӳɵIJ��A�Ľṹ��ʽΪ______________________��
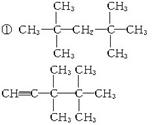
��4��ij�����л������������Է�������Ϊ88.0����C����������Ϊ68.2%����H����������Ϊ13.6%����������ײⶨ����һ���ǻ����˴Ź���������ʾ�÷�������3�������������ֲ�ͬ��������ԭ�ӣ���д����ṹ��ʽ_____________________.
��1����0.2 mol��A����������ȫȼ��ʱ����CO2��H2O��1.2 mol�������������2��2-�������飬��A�ĽṹʽΪ___________��
��2������ʽΪC6H12��ijϩ�������е�̼ԭ�Ӷ���ͬһƽ���ϣ����ϩ���Ľṹ��ʽΪ______________________��
��3��ij����A�����ܶ�����ͬ״���������ܶȵ�64�������ⶨ֪A�����й���6������
����A��������ϩ���������ӳɵIJ��A�Ľṹ��ʽΪ______________________��
����A��Ȳ���������ӳɵIJ��A�Ľṹ��ʽΪ______________________��
��4��ij�����л������������Է�������Ϊ88.0����C����������Ϊ68.2%����H����������Ϊ13.6%����������ײⶨ����һ���ǻ����˴Ź���������ʾ�÷�������3�������������ֲ�ͬ��������ԭ�ӣ���д����ṹ��ʽ_____________________.
|
|
 ��2��
��2��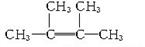
��3��
 ��4��
��4��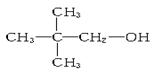
CXHY -----------xC-------YH
0.2mol 1.2mol 2.4mol ����ʽΪC6H12 (CH3)2CHCH(CH3)2
��2������ʽΪC6H12��ijϩ�������е�̼ԭ�Ӷ���ͬһƽ����Ϊ��ϩ�Ľṹ��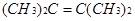
��3��ij����A�����ܶ�����ͬ״���������ܶȵ�64��Mr==128֪A�����й���6����
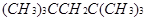 ��
��
��4��C:H:O==5:12:1��������ײⶨ����һ���ǻ����˴Ź���������ʾ�÷�������3�������������ֲ�ͬ��������ԭ��������һ���⣬�ǻ�һ���⣬����һ���⡣�ṹ��ʽΪ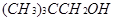
0.2mol 1.2mol 2.4mol ����ʽΪC6H12 (CH3)2CHCH(CH3)2
��2������ʽΪC6H12��ijϩ�������е�̼ԭ�Ӷ���ͬһƽ����Ϊ��ϩ�Ľṹ��
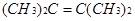
��3��ij����A�����ܶ�����ͬ״���������ܶȵ�64��Mr==128֪A�����й���6����
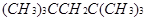 ��
��
��4��C:H:O==5:12:1��������ײⶨ����һ���ǻ����˴Ź���������ʾ�÷�������3�������������ֲ�ͬ��������ԭ��������һ���⣬�ǻ�һ���⣬����һ���⡣�ṹ��ʽΪ
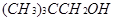

��ϰ��ϵ�д�
 ��ʦ����ɳ���ʱͬ��ѧ����ϵ�д�
��ʦ����ɳ���ʱͬ��ѧ����ϵ�д�
�����Ŀ
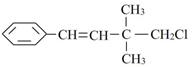
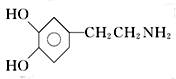
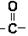 ������
������ ��ʾ�ķ���ʽ �������� ��
��ʾ�ķ���ʽ �������� ��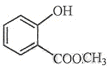 �к��еĹ����ŵ�����Ϊ
�к��еĹ����ŵ�����Ϊ 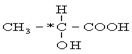
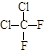 ��
��