��Ŀ����
������ĺ���������ҽҩ�������ȷ���������Ҫ����;��
��1��)�ؾ�ʯ��BaS04)����θ������Ӱ����
��֪�������£�Ksp(BaSO4)=1.1��10-10����BaSO4����Һ�м������ᣬ����Һ��pH=2ʱ�� ��Һ�� c(Ba2+)= ��
��2����������茶���[(NH4)2Fe(SO4)2��6H20]������������
�ټ��龧���к���NH4+�ķ���Ϊ_______��
�ڵ����ʵ���Ũ�ȵ�����ϡ��Һ��
a��(NH4)2Fe(SO4)2 b��NH4HSO4 c��(NH4)2SO4 d��(NH4)2SO3
����C(NH4+)�ɴ�С��˳��Ϊ ����ѡ����ĸ����
��3����������أ�K2S2O8)����ǿ��������Na2S2O3������ԭ����
��K2S2O8��Һ������MnSO4��Һ���,�ڴ��������£����Թ۲쵽��Һ��Ϊ��ɫ�� �÷�Ӧ�����ӷ���ʽΪ
���ò����缫�����H2SO4��K2SO4�Ļ����Һ�����Ʊ�K2S2O8���������ĵ缫��ӦʽΪ_____ __������������������Һ��pH��______ (���������С�����䡱)
�۲�Ʒ��K2S2O8�ĺ������õ������ⶨ����������Ϊ����ȡ0.3g��Ʒ�ڵ���ƿ�У���50 mLˮ�ܽ⣻����4.000 g KI����(�Թ���������ʹ���ַ�Ӧ����������������Һ�ữ����______Ϊָʾ������0.1000 mol��L-1 Na2S2O3��Һ�ζ����յ㣨��֪��I2��2S2O32��=2I����S4O62�������ظ�2�Σ����ƽ�����ı�Һ21.00mL���ò�Ʒ��K2S2O8����������Ϊ(���ʲ��μӷ�Ӧ) (��ʽ�����㣩��
��1��2.2��10��8 mol/L ��2�֣�
��2���� ȡ�������壬��ˮ�ܽ⣬�ټ�������NaOHŨ��Һ���ȣ�������ʹʪ��ĺ�ɫʯ����ֽ���������壬����NH4+��������������Ҳ���֣� ��2�֣�
�� a��c��d��b ��2�֣�д��a c d b ��b��d��c��aҲ�ɸ��֣���д��b d c a�����֣�
��3���� 5S2O82��+2Mn2++8H2O����10SO42��+2MnO4��+16H+ ��2�֣�
�� 2SO42�� ��2e��=S2O82������2HSO4�� ��2e�� = S2O82��+2H+�� ��2�֣�
���� ��1�֣�
�� ���� ��1�֣�
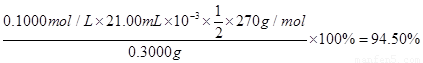
��3�֣���ʽ2�֣����1�֡���ʽ�в�����λ���۷֣�д��94.5%��۷֡���
��������
�����������1��pH=2ʱ��c(SO42-)=0.5��10-2mol/L��Ksp(BaSO4)=c(Ba2+)��c(SO42-)=1.1��10-10����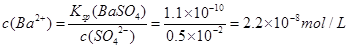 ��
��
��2����NH4+�ļ���ͨ���ǼӼʹ��ת��Ϊ��������ʪ���ʯ����ֽ���鰱����
��NH4+�����������ӣ���ˮ�п�ˮ��ʹ��Һ�����ԡ�a��NH4+��Fe2+��ˮ������ƣ�c��NH4+����ˮ�⣬d��NH4+��SO32-ˮ����ٽ������c(NH4+)�ɴ�СΪa>c>d����a��c��d�����ж���2��笠����ӣ���b������ֻ��1��笠����ӣ�ˮ��Ͼ������ģ����b�е�c(NH4+)��С��������˳��Ϊa��c��d��b��
��3������Һ����ɫ˵���и���������ɣ��������������ԭΪ�������
�ڵ��ʱ��������������ʧ���ӣ��ʵ缫��ӦʽΪ2SO42�� ��2e��=S2O82�������������ӵõ��ӣ�������ӦΪ2H++2e��=H2���������Ӽ�����Һ���Լ�����pH���ߡ�
�۸������������ζ�ԭ����Na2S2O3��Һ�����ζ��ⵥ�ʣ��������۱���������õ�����ָʾ����
�ζ�ʱ�����ķ�Ӧ�У�S2O82��+2I��=2SO42��+I2��I2��2S2O32��=2I����S4O62��������S2O82����2S2O32������˲�Ʒ��K2S2O8������Ϊ0.1000��21��10-3��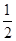 ��270=0.2835g����Ʒ��K2S2O8����������Ϊ
��270=0.2835g����Ʒ��K2S2O8����������Ϊ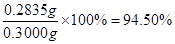 ��
��
���㣺�����ܶȻ��ļ��㣬�����ˮ�⣬���Ӽ��飬����ԭ�����к͵ζ�ԭ����

��1����ҵ����Na2SO3��Һ������ҵβ���е�SO2���������ݱ�ʾ��Ӧ������
| n(SO32-) |
| n(HSO3-) |
|
91��9 | 1��1 | 9��91 | ||
| ������pH | 8.2 | 7.2 | 6.2 |
| n(SO32-) |
| n(HSO3-) |
��2����֪Ki1��H2SO3����Ki��HAc����Ki2��H2SO3����Ki2��H2CO3����ҪʹNaHSO3��Һ��c��Na+����c��HSO3-���ӽ�1��1��������Һ�м�������
a��H2SO3��Һ b��NaOH��Һ c�������� d��Na2CO3
��3��ʵ����ͨ�����µ��KHSO4��Һ�Ʊ����������K2S2O8��д������KHSO4�ĵ��뷽��ʽ��
��4��S2O82-��ǿ�����ԣ���ԭ����ΪSO42-�������̣�MnSO4��������أ�K2S2O8����������Һ�������Ӵ��¿ɷ�����Ӧ���õ��Ϻ�ɫ��Һ����д�˷�Ӧ�Ļ�ѧ����ʽ��
��5����֪��S2O32-�н�ǿ�Ļ�ԭ�ԣ�ʵ���ҿ���I-�ⶨK2S2O8��Ʒ�Ĵ��ȣ���Ӧ����ʽΪ��
S2O82-+2I-��2SO42-+I2 ���٣�
I2+2S2O32-��2I-+S4O62-���ڣ�
��S2O82-��S4O62-��I2������ǿ��˳��
��6��K2S2O8��ƫ����ϩ��CH2=CF2���ۺϵ���������ƫ����ϩ��CH3-CClF2������ȥHCl�Ƶã�����0.5molƫ����ϩ����Ҫ����54kJ���ȣ�д����Ӧ���Ȼ�ѧ����ʽ