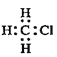��Ŀ����
����Ŀ����ԭ��Ϊԭ��������ϩ�����������£�
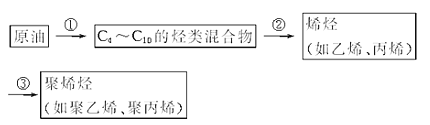
�ش��������⣺
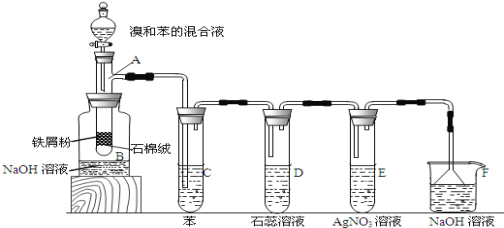
��1�������Тڱ�ʾ�ļӹ�������_____(�����)��
A������ B����Һ C���ѽ� D������
��2��C4��C10�Ļ�����к����飬��д�����������ͬ���칹��Ľṹ��ʽ��______��
��3������ϩ�Ľṹ��ʽΪ______��
��4����ϩ����ϩ��ѧ�������ơ���ϩ������ˮ��Ӧ���ɱ������÷�Ӧ������____�������й����ŵ�������____��
��5��д���Ҵ�������������ȩ�Ļ�ѧ����ʽ______��
���𰸡�C CH3-(CH2)3-CH3��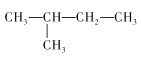 ��
��
![]() �ӳɷ�Ӧ �ǻ� 2CH3CH2OH+O2
�ӳɷ�Ӧ �ǻ� 2CH3CH2OH+O2![]() 2CH3CHO+2H2O
2CH3CHO+2H2O
��������
��1��������ͼ���Կ��������Тڱ�ʾ��������Է������ϴ�ĺ�C4��C10�����������ѳɷ�������С����ϩ����ϩ�ȣ����Դ��ּӹ��������ѽ⣬�ʴ�Ϊ��C��
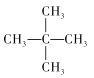
��2������ķ���ʽΪC5H12��������ͬ���칹�壬�ֱ�Ϊ�������飺CH3-(CH2)3-CH3�������飺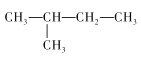 �������飺
�������飺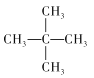 ���ʴ�Ϊ��CH3-(CH2)3-CH3��
���ʴ�Ϊ��CH3-(CH2)3-CH3��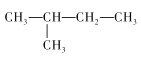 ��
�� ��
��
��3������ϩ�Ľṹ��ʽΪ��![]() ���ʴ�Ϊ��
���ʴ�Ϊ��![]() ��
��
��4���ɱ�ϩ��ˮ��Ӧ�Ʊ���Ϊ�ӳɷ�Ӧ�������к��еĹ�����Ϊ�ǻ����ʴ�Ϊ���ӳɷ�Ӧ���ǻ���
��5���Ҵ���ͭ�����������´�����������ȩ��ˮ����ѧ����ʽΪ��2CH3CH2OH+O2![]() 2CH3CHO+2H2O���ʴ�Ϊ��2CH3CH2OH+O2
2CH3CHO+2H2O���ʴ�Ϊ��2CH3CH2OH+O2![]() 2CH3CHO+2H2O��
2CH3CHO+2H2O��

 �����Ƹ���ʦ����ϵ�д�
�����Ƹ���ʦ����ϵ�д� ��ͨ����ͬ����ϰ��ϵ�д�
��ͨ����ͬ����ϰ��ϵ�д� ����С����ͬ������ϵ�д�
����С����ͬ������ϵ�д�