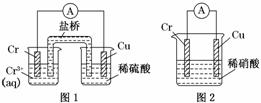
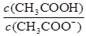��Ŀ����
�л���A���������Ƿ��͵õ���Ҳ���Դ�ţ������ȡ��������AΪ��ɫճ��Һ�壬������ˮ��Ϊ�о�A�������ṹ������������ʵ�飺
| ʵ�鲽�� | ���ͻ�ʵ����� |
| |
| (1)��ȡA 9.0 g������ʹ�������������ܶ�����ͬ������H2��45���� | ��ͨ��������գ�(1)A����Է�������Ϊ��____________�� |
| |
| (2)��9.0 g A��������O2�г��ȼ�գ���ʹ��������λ���ͨ��Ũ���ᡢ��ʯ�ң��������߷ֱ�����5.4 g��13.2 g�� | (2)A�ķ���ʽΪ��________________ _____________________________________________________�� | |
| (3)��ȡA 9.0 g����������NaHCO3��ĩ��Ӧ������2.24 LCO2(��״��)���������������Ʒ�Ӧ������2.24 L H2(��״��)�� | (3)�ýṹ��ʽ��ʾA�к��еĹ�����__________��________�� | |
| (4)A�ĺ˴Ź�����������ͼ��
| (4)A�к���______����ԭ�ӡ� | |
| (5)����������A�Ľṹ��ʽΪ________�� | ||
������(1)A���ܶ�����ͬ������H2�ܶȵ�45������Է�������Ϊ45��2��90��9.0 g A�����ʵ���Ϊ0.1 mol��
(2)ȼ�ղ������λ���ͨ��Ũ���ᡢ��ʯ�ң��������߷ֱ�����5.4 g��13.2 g��˵��0.1 mol Aȼ������0.3 mol H2O��0.3 mol CO2��1 mol A�к���6 mol H��3 mol C��
(3)0.1 mol A��������NaHCO3��ĩ��Ӧ������2.24 L CO2(��״��)���������������Ʒ�Ӧ������2.24 L H2(��״��)��˵�������к���һ����COOH��һ����OH��
(4)A�ĺ˴Ź�����������4���壬˵�������к���4�ִ��ڲ�ͬ��ѧ�����µ���ԭ�ӡ�
(5)����������A�Ľṹ��ʽΪ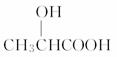 ��
��
�𰸡�(1)90��(2)C3H6O3��(3)��COOH����OH��(4)4��(5)

 Сѧ��ѧ������ѿڶ���ϵ�д�
Сѧ��ѧ������ѿڶ���ϵ�д� ������Ӧ�������������ϵ�д�
������Ӧ�������������ϵ�д� �㽭֮�ǿ�ʱ�Ż���ҵϵ�д�
�㽭֮�ǿ�ʱ�Ż���ҵϵ�д�

 ��OH��===CO
��OH��===CO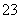 ��H2O
��H2O )��c(Cl��)��c(OH��)
)��c(Cl��)��c(OH��)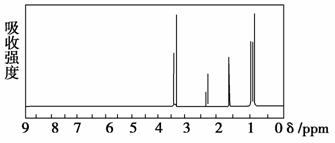
 D��CH3CH2CHO
D��CH3CH2CHO )��1,8����������(
)��1,8����������( )�Ļ������߿�����������������98%�����ᣬ��ǰ�߲��ܡ�������һ���ʿ��Խ�������ͬ���칹����룬�����������������������98%���ᣬ��ֽ��裬������©�����ˣ�������Һ�еõ�����1,8����������Ӧ���õķ�����
)�Ļ������߿�����������������98%�����ᣬ��ǰ�߲��ܡ�������һ���ʿ��Խ�������ͬ���칹����룬�����������������������98%���ᣬ��ֽ��裬������©�����ˣ�������Һ�еõ�����1,8����������Ӧ���õķ�����