��Ŀ����
��λ�ڢ�A�壬���Ȼ��� (BCl3)��������ȡ������(B2H6)��Ҳ�����л��ϳɵĴ��������Ȼ�����ˮ���ҷ�Ӧ��
�������ϣ���BCl3�ķе�Ϊ12.5 �棬�۵�Ϊ��107.3 �棻
��2B��6HCl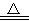 2BCl3����3H2����
2BCl3����3H2����
�����������������ƣ�Ҳ��������������Һ��Ӧ
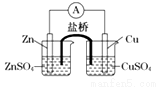
���ʵ�飺ijͬѧ�����ͼ��ʾװ�ã����������͵�����Ӧ�Ʊ����Ȼ���
�ش��������⣺
(1)д��Aװ���з�Ӧ�Ļ�ѧ����ʽ ��
(2)B������ ��C������ ��ʵ���������E���ռ����������� (�ѧʽ)
(3)G�����������պ�Cl2β����д��G�з�Ӧ�Ļ�ѧ����ʽ ��
(4)ʵ���п�����һ��ʢװ (���Լ�����)�ĸ���ܴ���F��Gװ�ã�ʹʵ�����㡣
(5)Ϊ��˳�����ʵ�飬��ȷ�IJ����� (�����)
���ȵ�ȼD���ƾ��ƣ����ȼA���ƾ���
���ȵ�ȼA���ƾ��ƣ����ȼD���ƾ���
��ͬʱ��ȼA��D���ƾ���
(6)���㲹����������ļ���ʵ�飬����֤�ƵõIJ�Ʒ���Ƿ�����ۣ�ȡ������Ʒ���Թ��У��μ�Ũ (�ѧʽ)��Һ���������ݲ���������Ʒ�к�����ۣ��������ݲ���������Ʒ������ۡ�
 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�25��ʱ��������ĵ���ƽ�ⳣ�����±���ʾ��
��ѧʽ | CH3COOH | HClO | H3PO3 |
���� | ���� | ������ | ������ |
����ƽ�ⳣ��(25��) | 1.8��10-5 | 3.0��10-8 | K1��8.3��10-3 K2��5.6��10-6 |
�ش��������⣺
��1�������ᣨH3PO3��Ϊ��Ԫ��ǿ�ᣬ���н�ǿ�Ļ�ԭ�ԣ��ɽ������ӻ�ԭ�������ʣ��ڿ����ɻ���������H3PO4��H3PO3�ĵ��뷽��ʽΪ ��
��2�����ʵ���Ũ�Ⱦ�Ϊ0.1 mol��L-1��������Һ��a��CH3COONa b��NaClO c��NaHPO3���ǵ�pH��С��������˳���� (����ĸ��ʾ)��
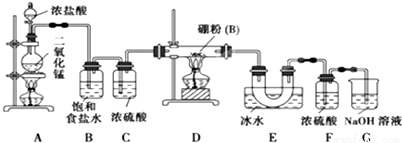
��3�������£�0.1 mol��L-1��CH3COOH��Һ��ˮϡ�����У����б���ʽ������һ����С����________������ĸ����
A��c��H+) B��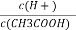
C��c(H+)��c(OH-) D��
��4�����Ϊ10 mL�� pH��Ϊ2�Ĵ�����Һ��һԪ��HX�ֱ��ˮϡ����1 000 mL��ϡ������pH�仯��ͼ��ʾ,��HX�ĵ���ƽ�ⳣ��________������ڡ��������ڡ���С�ڡ�)�����ƽ�ⳣ��


 2Z(g) һ���ﵽ��ѧƽ��״̬���ǣ�
2Z(g) һ���ﵽ��ѧƽ��״̬���ǣ�