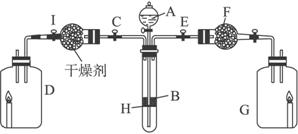
��Ŀ����
Ϊ��֤�й����ʵ����ʣ�ijѧ������С���������ͼ��ʾװ�ã�������C��ʹҺ��A����H�ϵĹ���B����ʱ���������������������D����������ɷ�ΪC20H42������Ϩ�𣻹ر�C��Eʱ����G������ȼ�ո�����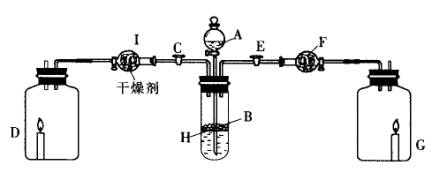
��1��д���������ʵ����ƣ�A________��B________��F________��
��2��I�и������ʹ�ã����ţ�________��
����ʯ������
����ʯ��
����ˮCaCl2 ��ŨH2SO4
��3��д�����л�ѧ����ʽ�������ӷ�Ӧ��ֻд���ӷ���ʽ��
��A��B�����壺______________________________________________________��
��F��ͨ�������壺____________________________________________________��
������
| ��1��A�����ᣬB��ʯ��ʯ��F���������ƣ���2������
��3����
|
��ʾ��
| ��Ϥ������Ļ�ѧ����
|

 ѧ�����νӽ̲��Ͼ���ѧ������ϵ�д�
ѧ�����νӽ̲��Ͼ���ѧ������ϵ�д� Сѧ������ҵϵ�д�
Сѧ������ҵϵ�д� ��ʿһ��ȫͨϵ�д�
��ʿһ��ȫͨϵ�д���15�֣�ij�о���ѧϰС�����ʵ��̽�����Ƚ��������ʣ�����Ƭ(��������Ĥ)Ͷ��Ũ�Ȼ�ͭ��Һ�У�������ܿ����һ�㺣��״����ɫ���ʣ���������Ƭ�ϲ����������ݡ���������֪��Һ�¶�Ѹ���������ռ����壬��������п�ȼ�ԡ�����ͬ������ƬͶ��ͬŨ�ȵ�����ͭ��Һ�У��ڶ�ʱ������Ƭ�����Ա仯��
�������Ȼ�ͭ��Һ��Ѹ�ٷ�Ӧ������ͬŨ�ȵ�����ͭ��Һ�ڶ�ʱ���ڲ���Ӧ��ԭ������� ��
A���Ȼ�ͭ��Һ���Ա�ͬŨ�ȵ�����ͭ��Һ����ǿ
B������ͭˮ����������ʹ���ۻ�
C�����������ƻ����������污Ĥ,����������Ӳ���
D�������Ȼ�������ˮ,������������������ˮ
�����һ����ʵ����֤���ѡ�� ��
����Ƭ������ֵİ���ɫ���ʵĹ����з��������ӷ�Ӧ����ʽ�� �������ӷ���ʽ���Ͳ������ݵ�ԭ�� ��
�Ƿų��������� ,����й����ʵ�Ũ�ȡ��������Ƿ��е绯ѧ���õȷ�����ʼ�β�����������ʲ��ϼӿ��ԭ���� ��(д��һ�㼴��)
��ijͬѧͨ��һ����ʵ�������Ҳ��ʹ��Ƭ������ͭ��Һ��Ӧ�ӿ죬����ȡ�Ĵ�ʩ������ ��
A����ɰֽ��ȥ��Ƭ���������Ĥ��Ͷ������ͭ��Һ��
B������ƬͶ��������������Һ��һ��ʱ���ȡ��ϴ�ӣ���Ͷ������ͭ��Һ��
C��������ͭ��Һ�еμ�����������Һ����Ͷ����Ƭ
D������Ƭ�����ϼ�öͭ����Ͷ��ͬŨ�ȵ�����ͭ��Һ��
�ɳ�ȥ����������Ƭ��ͭƬΪ�缫����X�������Һ�й���ԭ��أ��б����£�
| ѡ�� | ���缫 | ����� | ������Ӧ | ������Ӧ |
| A | ���� | NaOH | Al-3e-��Al3+ | 2H2O+2e-��2OH-+H2�� |
| B | ���� | ϡ���� | 2Al-6e-=2Al3+ | 6H++6e-��3H2�� |
| C | ���� | Ũ���� | Cu-2e--��Cu2+ | 2NO3-+4H+-4e-��2NO2��+2H2O |
| D | ���� | ϡ���� | Cu-2e-��Cu2+ | 2NO3-+8H+��2NO��+4H2O+6e- |
�� 15�֣�ij�о���ѧϰС�����ʵ��̽�����Ƚ��������ʣ�����Ƭ(��������Ĥ)Ͷ��Ũ�Ȼ�ͭ��Һ�У�������ܿ����һ�㺣��״����ɫ���ʣ���������Ƭ�ϲ����������ݡ���������֪��Һ�¶�Ѹ���������ռ����壬��������п�ȼ�ԡ�����ͬ������ƬͶ��ͬŨ�ȵ�����ͭ��Һ�У��ڶ�ʱ������Ƭ�����Ա仯��
�������Ȼ�ͭ��Һ��Ѹ�ٷ�Ӧ������ͬŨ�ȵ�����ͭ��Һ�ڶ�ʱ���ڲ���Ӧ��ԭ������� ��
A���Ȼ�ͭ��Һ���Ա�ͬŨ�ȵ�����ͭ��Һ����ǿ
B������ͭˮ����������ʹ���ۻ�
C�����������ƻ����������污Ĥ,����������Ӳ���
D�������Ȼ�������ˮ,������������������ˮ
�����һ����ʵ����֤���ѡ�� ��
����Ƭ������ֵİ���ɫ���ʵĹ����з��������ӷ�Ӧ����ʽ�� �� �����ӷ���ʽ���Ͳ������ݵ�ԭ�� ��
�Ƿų��������� ,����й����ʵ�Ũ�ȡ��������Ƿ��е绯ѧ���õȷ�����ʼ�β�����������ʲ��ϼӿ��ԭ���� ��(д��һ�㼴��)
��ijͬѧͨ��һ����ʵ�������Ҳ��ʹ��Ƭ������ͭ��Һ��Ӧ�ӿ죬����ȡ�Ĵ�ʩ������ ��
A����ɰֽ��ȥ��Ƭ���������Ĥ��Ͷ������ͭ��Һ��
B������ƬͶ��������������Һ��һ��ʱ���ȡ��ϴ�ӣ���Ͷ������ͭ��Һ��
C��������ͭ��Һ�еμ�����������Һ����Ͷ����Ƭ
D������Ƭ�����ϼ�öͭ����Ͷ��ͬŨ�ȵ�����ͭ��Һ��
�ɳ�ȥ����������Ƭ��ͭƬΪ�缫����X�������Һ�й���ԭ��أ��б����£�
|
ѡ�� |
���缫 |
����� |
������Ӧ |
������Ӧ |
|
A |
���� |
NaOH |
Al-3e-��Al3+ |
2H2O+2e-��2OH-+H2�� |
|
B |
���� |
ϡ���� |
2Al-6e-=2Al3+ |
6H++6e-��3H2�� |
|
C |
���� |
Ũ���� |
Cu-2e--��Cu2+ |
2NO3-+4H+-4e-��2NO2��+2H2O |
|
D |
���� |
ϡ���� |
Cu-2e-��Cu2+ |
2NO3-+8H+��2NO��+4H2O+6e- |
������ȷ���� ���ɴ���֪���������缫�����뱾��������أ������� ________ �йء�
 Al��OH��3+3H+��Cu2++2H2O
Al��OH��3+3H+��Cu2++2H2O Cu��OH��2+2H+��2Al+6H+=2Al3++3H2��
Cu��OH��2+2H+��2Al+6H+=2Al3++3H2��