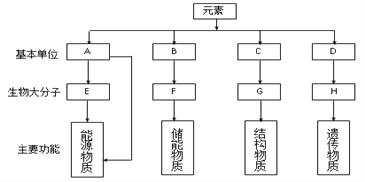
��Ŀ����
����Ŀ��ij�о�С������̽��Ӱ���������ֽ�����ص�����ʵ�顣��Ӧ��ʵ��������ͼ��ʾ��ʵ��1��ʵ��2�������������½�������������ش���
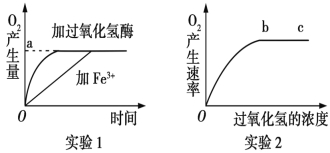
��1��ʵ��1��2�е��Ա����ֱ�Ϊ____________________________��
��2��ʵ��2�����ӳ��bc��O2�������ʲ��������ԭ���������_________________��
��3��ʵ��1���¶�������10�����ӹ�������ø�Ĵ���Ӧ����б�ʽ�_______�������С����;��Fe3+�Ĵ���Ӧ����б�ʽ�_______�������С������
���𰸡���1�����������ࡢ���������Ũ��
��2��ø����������
��3����С ����
����������1���۲���ͼ��֪ʵ��1��2���Ա����ֱ��Ǵ��������ࡢ���������Ũ�ȡ���2��ʵ��2��������bc��O2�������ʲ��������ԭ���������ø��������Ũ�������ޡ���3����֪ʵ�鶼�������������½��е�����ø�Ļ������¶ȵ�������Ӱ��������ʵ��1���¶�����10�����ӹ�������ø�Ĵ���Ӧ���ʽ���������б�ʽ���С����Fe3+�Ĵ���Ӧ��������������б�ʽ�����

 ��ͼͼ�麮����ҵ������ҵ���ִ�ѧ������ϵ�д�
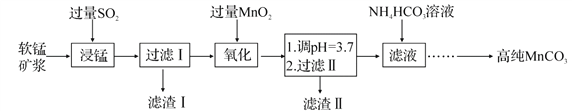
��ͼͼ�麮����ҵ������ҵ���ִ�ѧ������ϵ�д�����Ŀ����ҵ�����̿�(��Ҫ�ɷ��� MnO2������SiO2��Fe2O3����������)Ϊ��Ҫԭ���Ʊ������ܵĴ��Բ���̼����(MnCO3)���乤ҵ����������
��֪:25��ʱ,���ֽ���������������������ʽ����ʱ��Һ��pH ���±���ʾ��
���������� | Fe3+ | Fe2+ | Mn2+ |
��ʼ������pH | 2.7 | 7.6 | 8.3 |
��ȫ������pH | 3.7 | 9.7 | 9.8 |
(1)�������������� Fe2O3��SO2��Ӧ�����ӷ���ʽΪ Fe2O3+SO2+2H+ =2Fe2++SO42-+H2O���÷�Ӧ�Ǿ�������������Ӧʵ�ֵ���
д���������ӷ���ʽ��_______________________��
����Fe2O3+6H+ =2Fe3++3H2O ��������
(2)���ˢ�������Һ����Ҫ���ڵ����ֽ���������Ϊ_________ (�����ӷ���)��
(3)д����������������MnO2 ��Fe 2+��Ӧ�����ӷ���ʽ��___________��
(4)����������Ӧ�������и����� MnS2O6����,�¶ȶ�����������Ӧ��Ӱ����ͼ��ʾ��Ϊ����MnS2O6�����������������������¶�����_____������ijɷ���______ (д��ѧʽ)������ˢ����õ���Һ�м���NH4HCO3 ��Һʱ�¶Ȳ���̫�ߵ�ԭ����______________��
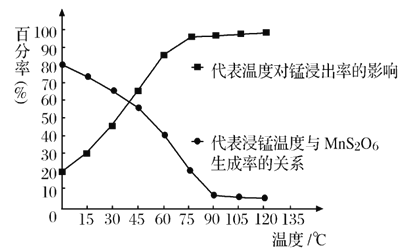
(5)����NH4HCO3��Һ��,���� MnCO3����,ͬʱ������������,д����Ӧ�����ӷ���ʽ��__________________ ��
(6)���ɵ� MnCO3�����辭���ϴ��������ϴ���Ƿ���ȫ�ķ�����_______________��