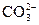��Ŀ����
�������ӷ���ʽ��д����ȷ���ǣ� ��
| A��Cl2����NaOH��Һ��Ӧ��Cl2+2OH��= Cl��+ClO��+ H2O |
| B����Ba(OH)2ϡ��Һ�еμ�NH4HSO4ϡ��Һ��Ba2���պó�����ȫ��Ba2����2OH����NH4����H����SO42��==BaSO4����NH3��H2O��H2O |
| C������������Һ��ϡ���ᡢ˫��ˮ��ϣ�2Fe2����H2O2��2H��==2Fe3����2H2O |
| D����������Һ�м��������ˮ��Al3����4NH3��H2O==AlO2����4NH4����2H2O |
D
�𰸣�D
A����ȷ��B����ȷ����֤Ba2����SO42�D�ı���Ϊ1��1��C����ȷ��˫��ˮ������������D����ȷ��NH3��H2O����������ܽ�Al (OH)3 ,Al3����3NH3��H2O=Al(OH)3����3NH4����

��ϰ��ϵ�д�
�����Ŀ

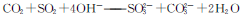
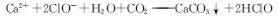
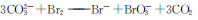
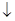 +2HClO
+2HClO Al(OH)3��
Al(OH)3�� H2��+C12��
H2��+C12��