��Ŀ����
����Ŀ�������ᣨ��ʽ��ϩ���ᣩ��Fe2+�γɵ�������������������ֳ�����Ѫ����������������ȱ����ƶѪ�������Ǻϳɸ���������һ�ֹ���·�ߣ�
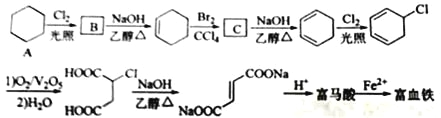
�ش��������⣺
��1��A�Ļ�ѧ����Ϊ_____����A����B�ķ�Ӧ����Ϊ_____��
��2��C�Ľṹ��ʽΪ_____��
��3��������Ľṹ��ʽΪ_____��
��4�����鸻Ѫ�����Ƿ���Fe3+��ʵ�����������_____��
��5��������Ϊ��Ԫ���ᣬ1mol����������������NaHCO3��Һ��Ӧ�ɷų�____L CO2����������������ͬ���칹���У�ͬΪ��Ԫ����Ļ���____________��д���ṹ��ʽ����
���𰸡���1��������ȡ����Ӧ��ÿ��2�֣���4�֣�
��2��![]() ��2�֣�
��2�֣�
��3��![]() ��2�֣�
��2�֣�
��4��ȡ������Ѫ��������ϡ�����ܽ⣬�ٵμ�KSCN��Һ������Һ��Ѫ��ɫ�����Ʒ�к���Fe3+����֮�����ޣ�2�֣�
��5��44.8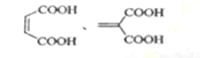 ��ÿ��2�֣���4�֣�
��ÿ��2�֣���4�֣�
�������������������1��A�Ļ�ѧ����Ϊ�����飬�ɻ������ڹ�������������������ȡ����Ӧ������1-�Ȼ����顣
��2������ϩ��������Ȼ�̼��Һ�����ӳɷ�Ӧ����C��C�Ľṹ��ʽΪ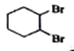 ��
��
��3������ϩ�����ữ�ø����ᣬ��ṹ��ʽΪ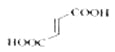 ��
��
��4������Fe3���ļ��顣���鸻Ѫ�����Ƿ���Fe3+��ʵ�����������ȡ������Ѫ��������ϡ�����ܽ⣬�ٵμ�KSCN��Һ������Һ��Ѫ��ɫ�����Ʒ�к���Fe3+����֮�����ޡ�
��5��������Ϊ��Ԫ���ᣬ1mol����������������NaHCO3��Һ��Ӧ����2molCO2����״�������Ϊ44.8LCO2���������ͬ���칹���У�ͬΪ��Ԫ����Ļ���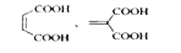 ��
��

 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�����Ŀ�������±������ļ������ʵ��۵㡢�е������ж�˵���д�������� ��
���� | NaCl | MgCl2 | AlCl3 | SiCl4 | ����R |
�۵� | 810�� | 710�� | 180�� | -70�� | 2300�� |
�е� | 1465�� | 1418�� | 177.8�� | 57�� | 2500�� |
A��AlCl3Ϊ���Ӿ���
B��MgCl2Ϊ���Ӿ���
C��SiCl4�Ƿ��Ӿ���
D������R������ԭ�Ӿ���